+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8ke9 | ||||||
|---|---|---|---|---|---|---|---|
| Title | The CBD domain of cyanophage A-1(L) short tail fiber | ||||||
 Components Components | Tail fiber protein | ||||||
 Keywords Keywords |  VIRAL PROTEIN / VIRAL PROTEIN /  fiber / fiber /  virus virus | ||||||
| Biological species |  unclassified Caudoviricetes (virus) unclassified Caudoviricetes (virus) | ||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.76 Å cryo EM / Resolution: 3.76 Å | ||||||
 Authors Authors | Yu, R.C. / Li, Q. / Zhou, C.Z. | ||||||
| Funding support |  China, 1items China, 1items
| ||||||
 Citation Citation |  Journal: Nat Commun / Year: 2024 Journal: Nat Commun / Year: 2024Title: Structure of the intact tail machine of Anabaena myophage A-1(L). Authors: Rong-Cheng Yu / Feng Yang / Hong-Yan Zhang / Pu Hou / Kang Du / Jie Zhu / Ning Cui / Xudong Xu / Yuxing Chen / Qiong Li / Cong-Zhao Zhou /  Abstract: The Myoviridae cyanophage A-1(L) specifically infects the model cyanobacteria Anabaena sp. PCC 7120. Following our recent report on the capsid structure of A-1(L), here we present the high-resolution ...The Myoviridae cyanophage A-1(L) specifically infects the model cyanobacteria Anabaena sp. PCC 7120. Following our recent report on the capsid structure of A-1(L), here we present the high-resolution cryo-EM structure of its intact tail machine including the neck, tail and attached fibers. Besides the dodecameric portal, the neck contains a canonical hexamer connected to a unique pentadecamer that anchors five extended bead-chain-like neck fibers. The 1045-Å-long contractile tail is composed of a helical bundle of tape measure proteins surrounded by a layer of tube proteins and a layer of sheath proteins, ended with a five-component baseplate. The six long and six short tail fibers are folded back pairwise, each with one end anchoring to the baseplate and the distal end pointing to the capsid. Structural analysis combined with biochemical assays further enable us to identify the dual hydrolytic activities of the baseplate hub, in addition to two host receptor binding domains in the tail fibers. Moreover, the structure of the intact A-1(L) also helps us to reannotate its genome. These findings will facilitate the application of A-1(L) as a chassis cyanophage in synthetic biology. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8ke9.cif.gz 8ke9.cif.gz | 126.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8ke9.ent.gz pdb8ke9.ent.gz | 95.1 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8ke9.json.gz 8ke9.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ke/8ke9 https://data.pdbj.org/pub/pdb/validation_reports/ke/8ke9 ftp://data.pdbj.org/pub/pdb/validation_reports/ke/8ke9 ftp://data.pdbj.org/pub/pdb/validation_reports/ke/8ke9 | HTTPS FTP |
|---|
-Related structure data
| Related structure data | 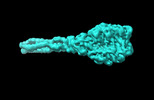 37150MC  8keaC  8kecC  8keeC  8kefC  8kegC  8ts6C M: map data used to model this data C: citing same article ( |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
| #1: Protein | Mass: 49010.348 Da / Num. of mol.: 3 / Source method: isolated from a natural source / Source: (natural)  unclassified Caudoviricetes (virus) unclassified Caudoviricetes (virus) |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component | Name: unclassified Caudoviricetes / Type: VIRUS / Entity ID: all / Source: NATURAL |
|---|---|
| Source (natural) | Organism:  unclassified Caudoviricetes (virus) unclassified Caudoviricetes (virus) |
| Details of virus | Empty: NO / Enveloped: NO / Isolate: OTHER / Type: VIRION |
| Natural host | Organism: Nostoc sp. PCC 7120 = FACHB-418 |
| Buffer solution | pH: 7.5 |
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YES |
Vitrification | Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 300 K |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 2500 nm / Nominal defocus min: 1500 nm Bright-field microscopy / Nominal defocus max: 2500 nm / Nominal defocus min: 1500 nm |
| Image recording | Electron dose: 50 e/Å2 / Film or detector model: GATAN K2 SUMMIT (4k x 4k) |
- Processing
Processing
| EM software | Name: RELION / Version: 3.1 / Category: 3D reconstruction |
|---|---|
CTF correction | Type: PHASE FLIPPING ONLY |
3D reconstruction | Resolution: 3.76 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 246372 / Symmetry type: POINT |
 Movie
Movie Controller
Controller






 PDBj
PDBj