+ Open data
Open data
- Basic information
Basic information
| Entry |  | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cytoplasmic dynein-1 heavy chain bound to JIP3-LZI | ||||||||||||
 Map data Map data | |||||||||||||
 Sample Sample |
| ||||||||||||
 Keywords Keywords |  Dynein / Dynein /  AAA-Atpase / JIP3 / AAA-Atpase / JIP3 /  MOTOR PROTEIN MOTOR PROTEIN | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationtransport along microtubule / dynein light chain binding / dynein heavy chain binding / positive regulation of intracellular transport / regulation of metaphase plate congression / anterograde axonal protein transport / establishment of spindle localization / positive regulation of spindle assembly / MAP-kinase scaffold activity /  P-body assembly ...transport along microtubule / dynein light chain binding / dynein heavy chain binding / positive regulation of intracellular transport / regulation of metaphase plate congression / anterograde axonal protein transport / establishment of spindle localization / positive regulation of spindle assembly / MAP-kinase scaffold activity / P-body assembly ...transport along microtubule / dynein light chain binding / dynein heavy chain binding / positive regulation of intracellular transport / regulation of metaphase plate congression / anterograde axonal protein transport / establishment of spindle localization / positive regulation of spindle assembly / MAP-kinase scaffold activity /  P-body assembly / P-body assembly /  dynein complex / COPI-independent Golgi-to-ER retrograde traffic / minus-end-directed microtubule motor activity / dynein complex / COPI-independent Golgi-to-ER retrograde traffic / minus-end-directed microtubule motor activity /  cytoplasmic dynein complex / JUN kinase binding / retrograde axonal transport / dynein light intermediate chain binding / nuclear migration / axon regeneration / centrosome localization / cytoplasmic dynein complex / JUN kinase binding / retrograde axonal transport / dynein light intermediate chain binding / nuclear migration / axon regeneration / centrosome localization /  microtubule motor activity / dynein intermediate chain binding / axon development / microtubule-based movement / microtubule motor activity / dynein intermediate chain binding / axon development / microtubule-based movement /  kinesin binding / regulation of JNK cascade / cytoplasmic microtubule / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / COPI-mediated anterograde transport / kinesin binding / regulation of JNK cascade / cytoplasmic microtubule / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / COPI-mediated anterograde transport /  stress granule assembly / cytoplasmic microtubule organization / Mitotic Prometaphase / regulation of mitotic spindle organization / EML4 and NUDC in mitotic spindle formation / axon cytoplasm / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / vesicle-mediated transport / Recruitment of mitotic centrosome proteins and complexes / Resolution of Sister Chromatid Cohesion / Recruitment of NuMA to mitotic centrosomes / HSP90 chaperone cycle for steroid hormone receptors (SHR) in the presence of ligand / Anchoring of the basal body to the plasma membrane / MHC class II antigen presentation / AURKA Activation by TPX2 / cellular response to nerve growth factor stimulus / mitotic spindle organization / stress granule assembly / cytoplasmic microtubule organization / Mitotic Prometaphase / regulation of mitotic spindle organization / EML4 and NUDC in mitotic spindle formation / axon cytoplasm / Loss of Nlp from mitotic centrosomes / Loss of proteins required for interphase microtubule organization from the centrosome / vesicle-mediated transport / Recruitment of mitotic centrosome proteins and complexes / Resolution of Sister Chromatid Cohesion / Recruitment of NuMA to mitotic centrosomes / HSP90 chaperone cycle for steroid hormone receptors (SHR) in the presence of ligand / Anchoring of the basal body to the plasma membrane / MHC class II antigen presentation / AURKA Activation by TPX2 / cellular response to nerve growth factor stimulus / mitotic spindle organization /  filopodium / RHO GTPases Activate Formins / positive regulation of JNK cascade / microtubule cytoskeleton organization / filopodium / RHO GTPases Activate Formins / positive regulation of JNK cascade / microtubule cytoskeleton organization /  kinetochore / Aggrephagy / HCMV Early Events / Separation of Sister Chromatids / azurophil granule lumen / kinetochore / Aggrephagy / HCMV Early Events / Separation of Sister Chromatids / azurophil granule lumen /  Regulation of PLK1 Activity at G2/M Transition / signaling receptor complex adaptor activity / late endosome / Regulation of PLK1 Activity at G2/M Transition / signaling receptor complex adaptor activity / late endosome /  cell body / positive regulation of cold-induced thermogenesis / cell body / positive regulation of cold-induced thermogenesis /  cell cortex / cell cortex /  growth cone / cytoplasmic vesicle / vesicle / growth cone / cytoplasmic vesicle / vesicle /  microtubule / protein stabilization / microtubule / protein stabilization /  cell division / cell division /  axon / axon /  Golgi membrane / Golgi membrane /  centrosome / centrosome /  dendrite / Neutrophil degranulation / negative regulation of apoptotic process / perinuclear region of cytoplasm / dendrite / Neutrophil degranulation / negative regulation of apoptotic process / perinuclear region of cytoplasm /  ATP hydrolysis activity / ATP hydrolysis activity /  RNA binding / extracellular exosome / extracellular region / RNA binding / extracellular exosome / extracellular region /  ATP binding / ATP binding /  membrane / identical protein binding / membrane / identical protein binding /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | ||||||||||||
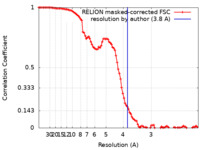
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.8 Å cryo EM / Resolution: 3.8 Å | ||||||||||||
 Authors Authors | Singh K / Lau CK / Manigrasso G / Gassmann R / Carter AP | ||||||||||||
| Funding support |  United Kingdom, European Union, 3 items United Kingdom, European Union, 3 items
| ||||||||||||
 Citation Citation |  Journal: Science / Year: 2024 Journal: Science / Year: 2024Title: Molecular mechanism of dynein-dynactin complex assembly by LIS1. Authors: Kashish Singh / Clinton K Lau / Giulia Manigrasso / José B Gama / Reto Gassmann / Andrew P Carter /   Abstract: Cytoplasmic dynein is a microtubule motor vital for cellular organization and division. It functions as a ~4-megadalton complex containing its cofactor dynactin and a cargo-specific coiled-coil ...Cytoplasmic dynein is a microtubule motor vital for cellular organization and division. It functions as a ~4-megadalton complex containing its cofactor dynactin and a cargo-specific coiled-coil adaptor. However, how dynein and dynactin recognize diverse adaptors, how they interact with each other during complex formation, and the role of critical regulators such as lissencephaly-1 (LIS1) protein (LIS1) remain unclear. In this study, we determined the cryo-electron microscopy structure of dynein-dynactin on microtubules with LIS1 and the lysosomal adaptor JIP3. This structure reveals the molecular basis of interactions occurring during dynein activation. We show how JIP3 activates dynein despite its atypical architecture. Unexpectedly, LIS1 binds dynactin's p150 subunit, tethering it along the length of dynein. Our data suggest that LIS1 and p150 constrain dynein-dynactin to ensure efficient complex formation. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_17832.map.gz emd_17832.map.gz | 75.6 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-17832-v30.xml emd-17832-v30.xml emd-17832.xml emd-17832.xml | 25.6 KB 25.6 KB | Display Display |  EMDB header EMDB header |
| FSC (resolution estimation) |  emd_17832_fsc.xml emd_17832_fsc.xml | 10 KB | Display |  FSC data file FSC data file |
| Images |  emd_17832.png emd_17832.png | 89.1 KB | ||
| Masks |  emd_17832_msk_1.map emd_17832_msk_1.map | 83.7 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-17832.cif.gz emd-17832.cif.gz | 9.9 KB | ||
| Others |  emd_17832_additional_1.map.gz emd_17832_additional_1.map.gz emd_17832_half_map_1.map.gz emd_17832_half_map_1.map.gz emd_17832_half_map_2.map.gz emd_17832_half_map_2.map.gz | 65 MB 65.2 MB 65.3 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-17832 http://ftp.pdbj.org/pub/emdb/structures/EMD-17832 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17832 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-17832 | HTTPS FTP |
-Related structure data
| Related structure data |  8pr2MC  8pqvC  8pqwC  8pqyC  8pqzC  8pr0C  8pr1C  8pr3C  8pr4C  8pr5C  8ptkC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_17832.map.gz / Format: CCP4 / Size: 83.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_17832.map.gz / Format: CCP4 / Size: 83.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.059 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_17832_msk_1.map emd_17832_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
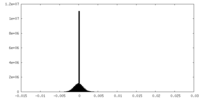
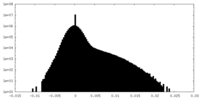
| Density Histograms |
-Additional map: unsharpened map
| File | emd_17832_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | unsharpened map | ||||||||||||
| Projections & Slices |
| ||||||||||||
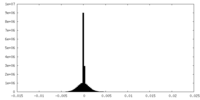
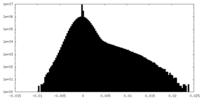
| Density Histograms |
-Half map: #2
| File | emd_17832_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_17832_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Cytoplasmic dynein-A heavy chain bound to dynactin p150 and IC-LC...
| Entire | Name: Cytoplasmic dynein-A heavy chain bound to dynactin p150 and IC-LC tower |
|---|---|
| Components |
|
-Supramolecule #1: Cytoplasmic dynein-A heavy chain bound to dynactin p150 and IC-LC...
| Supramolecule | Name: Cytoplasmic dynein-A heavy chain bound to dynactin p150 and IC-LC tower type: complex / ID: 1 / Parent: 0 / Macromolecule list: all |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: C-Jun-amino-terminal kinase-interacting protein 3
| Macromolecule | Name: C-Jun-amino-terminal kinase-interacting protein 3 / type: protein_or_peptide / ID: 1 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 65.975398 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: SNIEFLKMME IQMDEGGGVV VYQDDYCSGS VMSERVSGLA GSIYREFERL IHCYDEEVVK ELMPLVVNVL ENLDSVLSEN QEHEVELEL LREDNEQLLT QYEREKALRR QAEEKFIEFE DALEQEKKEL QIQVEHYEFQ TRQLELKAKN YADQISRLEE R ESEMKKEY ...String: SNIEFLKMME IQMDEGGGVV VYQDDYCSGS VMSERVSGLA GSIYREFERL IHCYDEEVVK ELMPLVVNVL ENLDSVLSEN QEHEVELEL LREDNEQLLT QYEREKALRR QAEEKFIEFE DALEQEKKEL QIQVEHYEFQ TRQLELKAKN YADQISRLEE R ESEMKKEY NALHQRHTEM IQTYVEHIER SKMQQVGGNS QTESSLPGRR KERPTSLNVF PLADGTVRAQ IGGKLVPAGD HW HLSDLGQ LQSSSSYQCP QDEMSESGQS SAAATPSTTG TKSNTPTSSV PSAAVTPLNE SLQPLGDYGV GSKNSKRARE KRD SRNMEV QVTQEMRNVS IGMGSSDEWS DVQDIIDSTP ELDMCPETRL DRTGSSPTQG IVNKAFGINT DSLYHELSTA GSEV IGDVD EGADLLGEFS VRDDFFGMGK EVGNLLLENS QLLETKNALN VVKNDLIAKV DQLSGEQEVL RGELEAAKQA KVKLE NRIK ELEEELKRVK SEAIIARREP KEEAEDVSSY LCTESDKIPM AQRRRFTRVE MARVLMERNQ YKERLMELQE AVRWTE MIR ASREGSGSGR WSHPQFEK UniProtKB: C-Jun-amino-terminal kinase-interacting protein 3 |
-Macromolecule #2: Cytoplasmic dynein 1 heavy chain 1
| Macromolecule | Name: Cytoplasmic dynein 1 heavy chain 1 / type: protein_or_peptide / ID: 2 / Number of copies: 2 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 533.055125 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MSEPGGGGGE DGSAGLEVSA VQNVADVSVL QKHLRKLVPL LLEDGGEAPA ALEAALEEKS ALEQMRKFLS DPQVHTVLVE RSTLKEDVG DEGEEEKEFI SYNINIDIHY GVKSNSLAFI KRTPVIDADK PVSSQLRVLT LSEDSPYETL HSFISNAVAP F FKSYIRES ...String: MSEPGGGGGE DGSAGLEVSA VQNVADVSVL QKHLRKLVPL LLEDGGEAPA ALEAALEEKS ALEQMRKFLS DPQVHTVLVE RSTLKEDVG DEGEEEKEFI SYNINIDIHY GVKSNSLAFI KRTPVIDADK PVSSQLRVLT LSEDSPYETL HSFISNAVAP F FKSYIRES GKADRDGDKM APSVEKKIAE LEMGLLHLQQ NIEIPEISLP IHPMITNVAK QCYERGEKPK VTDFGDKVED PT FLNQLQS GVNRWIREIQ KVTKLDRDPA SGTALQEISF WLNLERALYR IQEKRESPEV LLTLDILKHG KRFHATVSFD TDT GLKQAL ETVNDYNPLM KDFPLNDLLS ATELDKIRQA LVAIFTHLRK IRNTKYPIQR ALRLVEAISR DLSSQLLKVL GTRK LMHVA YEEFEKVMVA CFEVFQTWDD EYEKLQVLLR DIVKRKREEN LKMVWRINPA HRKLQARLDQ MRKFRRQHEQ LRAVI VRVL RPQVTAVAQQ NQGEVPEPQD MKVAEVLFDA ADANAIEEVN LAYENVKEVD GLDVSKEGTE AWEAAMKRYD ERIDRV ETR ITARLRDQLG TAKNANEMFR IFSRFNALFV RPHIRGAIRE YQTQLIQRVK DDIESLHDKF KVQYPQSQAC KMSHVRD LP PVSGSIIWAK QIDRQLTAYM KRVEDVLGKG WENHVEGQKL KQDGDSFRMK LNTQEIFDDW ARKVQQRNLG VSGRIFTI E STRVRGRTGN VLKLKVNFLP EIITLSKEVR NLKWLGFRVP LAIVNKAHQA NQLYPFAISL IESVRTYERT CEKVEERNT ISLLVAGLKK EVQALIAEGI ALVWESYKLD PYVQRLAETV FNFQEKVDDL LIIEEKIDLE VRSLETCMYD HKTFSEILNR VQKAVDDLN LHSYSNLPIW VNKLDMEIER ILGVRLQAGL RAWTQVLLGQ AEDKAEVDMD TDAPQVSHKP GGEPKIKNVV H ELRITNQV IYLNPPIEEC RYKLYQEMFA WKMVVLSLPR IQSQRYQVGV HYELTEEEKF YRNALTRMPD GPVALEESYS AV MGIVSEV EQYVKVWLQY QCLWDMQAEN IYNRLGEDLN KWQALLVQIR KARGTFDNAE TKKEFGPVVI DYGKVQSKVN LKY DSWHKE VLSKFGQMLG SNMTEFHSQI SKSRQELEQH SVDTASTSDA VTFITYVQSL KRKIKQFEKQ VELYRNGQRL LEKQ RFQFP PSWLYIDNIE GEWGAFNDIM RRKDSAIQQQ VANLQMKIVQ EDRAVESRTT DLLTDWEKTK PVTGNLRPEE ALQAL TIYE GKFGRLKDDR EKCAKAKEAL ELTDTGLLSG SEERVQVALE ELQDLKGVWS ELSKVWEQID QMKEQPWVSV QPRKLR QNL DALLNQLKSF PARLRQYASY EFVQRLLKGY MKINMLVIEL KSEALKDRHW KQLMKRLHVN WVVSELTLGQ IWDVDLQ KN EAIVKDVLLV AQGEMALEEF LKQIREVWNT YELDLVNYQN KCRLIRGWDD LFNKVKEHIN SVSAMKLSPY YKVFEEDA L SWEDKLNRIM ALFDVWIDVQ RRWVYLEGIF TGSADIKHLL PVETQEFQSI STEFLALMKK VSKSPLVMDV LNIQGVQRS LERLADLLGE IQKALGEYLE RERSSFPRFY FVGDEDLLEI IGNSKNVAKL QKHFKKMFAG VSSIILNEDN SVVLGISSRE GEEVMFKTP VSITEHPKIN EWLTLVEKEM RVTLAKLLAE SVTEVEIFGK ATSIDPNTYI TWIDKYQAQL VVLSAQIAWS E NVETALSS MGGGGDAAPL HSVLSNVEVT LNVLADSVLM EQPPLRRRKL EHLITELVHQ RDVTRSLIKS KIDNAKSFEW LS QMRFYFD PKQTDVLQQL SIQMANAKFN YGFEYLGVQD KLVQTPLTDR CYLTMTQALE ARLGGSPFGP AGTGKTESVK ALG HQLGRF VLVFNCDETF DFQAMGRIFV GLCQVGAWGC FDEFNRLEER MLSAVSQQVQ CIQEALREHS NPNYDKTSAP ITCE LLNKQ VKVSPDMAIF ITMNPGYAGR SNLPDNLKKL FRSLAMTKPD RQLIAQVMLY SQGFRTAEVL ANKIVPFFKL CDEQL SSQS HYDFGLRALK SVLVSAGNVK RERIQKIKRE KEERGEAVDE GEIAENLPEQ EILIQSVCET MVPKLVAEDI PLLFSL LSD VFPGVQYHRG EMTALREELK KVCQEMYLTY GDGEEVGGMW VEKVLQLYQI TQINHGLMMV GPSGSGKSMA WRVLLKA LE RLEGVEGVAH IIDPKAISKD HLYGTLDPNT REWTDGLFTH VLRKIIDSVR GELQKRQWIV FDGDVDPEWV ENLNSVLD D NKLLTLPNGE RLSLPPNVRI MFEVQDLKYA TLATVSRCGM VWFSEDVLST DMIFNNFLAR LRSIPLDEGE DEAQRRRKG KEDEGEEAAS PMLQIQRDAA TIMQPYFTSN GLVTKALEHA FQLEHIMDLT RLRCLGSLFS MLHQACRNVA QYNANHPDFP MQIEQLERY IQRYLVYAIL WSLSGDSRLK MRAELGEYIR RITTVPLPTA PNIPIIDYEV SISGEWSPWQ AKVPQIEVET H KVAAPDVV VPTLDTVRHE ALLYTWLAEH KPLVLCGPPG SGKTMTLFSA LRALPDMEVV GLNFSSATTP ELLLKTFDHY CE YRRTPNG VVLAPVQLGK WLVLFCDEIN LPDMDKYGTQ RVISFIRQMV EHGGFYRTSD QTWVKLERIQ FVGACNPPTD PGR KPLSHR FLRHVPVVYV DYPGPASLTQ IYGTFNRAML RLIPSLRTYA EPLTAAMVEF YTMSQERFTQ DTQPHYIYSP REMT RWVRG IFEALRPLET LPVEGLIRIW AHEALRLFQD RLVEDEERRW TDENIDTVAL KHFPNIDREK AMSRPILYSN WLSKD YIPV DQEELRDYVK ARLKVFYEEE LDVPLVLFNE VLDHVLRIDR IFRQPQGHLL LIGVSGAGKT TLSRFVAWMN GLSVYQ IKV HRKYTGEDFD EDLRTVLRRS GCKNEKIAFI MDESNVLDSG FLERMNTLLA NGEVPGLFEG DEYATLMTQC KEGAQKE GL MLDSHEELYK WFTSQVIRNL HVVFTMNPSS EGLKDRAATS PALFNRCVLN WFGDWSTEAL YQVGKEFTSK MDLEKPNY I VPDYMPVVYD KLPQPPSHRE AIVNSCVFVH QTLHQANARL AKRGGRTMAI TPRHYLDFIN HYANLFHEKR SELEEQQMH LNVGLRKIKE TVDQVEELRR DLRIKSQELE VKNAAANDKL KKMVKDQQEA EKKKVMSQEI QEQLHKQQEV IADKQMSVKE DLDKVEPAV IEAQNAVKSI KKQHLVEVRS MANPPAAVKL ALESICLLLG ESTTDWKQIR SIIMRENFIP TIVNFSAEEI S DAIREKMK KNYMSNPSYN YEIVNRASLA CGPMVKWAIA QLNYADMLKR VEPLRNELQK LEDDAKDNQQ KANEVEQMIR DL EASIARY KEEYAVLISE AQAIKADLAA VEAKVNRSTA LLKSLSAERE RWEKTSETFK NQMSTIAGDC LLSAAFIAYA GYF DQQMRQ NLFTTWSHHL QQANIQFRTD IARTEYLSNA DERLRWQASS LPADDLCTEN AIMLKRFNRY PLIIDPSGQA TEFI MNEYK DRKITRTSFL DDAFRKNLES ALRFGNPLLV QDVESYDPVL NPVLNREVRR TGGRVLITLG DQDIDLSPSF VIFLS TRDP TVEFPPDLCS RVTFVNFTVT RSSLQSQCLN EVLKAERPDV DEKRSDLLKL QGEFQLRLRQ LEKSLLQALN EVKGRI LDD DTIITTLENL KREAAEVTRK VEETDIVMQE VETVSQQYLP LSTACSSIYF TMESLKQIHF LYQYSLQFFL DIYHNVL YE NPNLKGVTDH TQRLSIITKD LFQVAFNRVA RGMLHQDHIT FAMLLARIKL KGTVGEPTYD AEFQHFLRGN EIVLSAGS T PRIQGLTVEQ AEAVVRLSCL PAFKDLIAKV QADEQFGIWL DSSSPEQTVP YLWSEETPAT PIGQAIHRLL LIQAFRPDR LLAMAHMFVS TNLGESFMSI MEQPLDLTHI VGTEVKPNTP VLMCSVPGYD ASGHVEDLAA EQNTQITSIA IGSAEGFNQA DKAINTAVK SGRWVMLKNV HLAPGWLMQL EKKLHSLQPH ACFRLFLTME INPKVPVNLL RAGRIFVFEP PPGVKANMLR T FSSIPVSR ICKSPNERAR LYFLLAWFHA IIQERLRYAP LGWSKKYEFG ESDLRSACDT VDTWLDDTAK GRQNISPDKI PW SALKTLM AQSIYGGRVD NEFDQRLLNT FLERLFTTRS FDSEFKLACK VDGHKDIQMP DGIRREEFVQ WVELLPDTQT PSW LGLPNN AERVLLTTQG VDMISKMLKM QMLEDEDDLA YAETEKKTRT DSTSDGRPAW MRTLHTTASN WLHLIPQTLS HLKR TVENI KDPLFRFFER EVKMGAKLLQ DVRQDLADVV QVCEGKKKQT NYLRTLINEL VKGILPRSWS HYTVPAGMTV IQWVS DFSE RIKQLQNISL AAASGGAKEL KNIHVCLGGL FVPEAYITAT RQYVAQANSW SLEELCLEVN VTTSQGATLD ACSFGV TGL KLQGATCNNN KLSLSNAIST ALPLTQLRWV KQTNTEKKAS VVTLPVYLNF TRADLIFTVD FEIATKEDPR SFYERGV AV LCTE UniProtKB: Cytoplasmic dynein 1 heavy chain 1 |
-Macromolecule #3: Cytoplasmic dynein 1 intermediate chain 2
| Macromolecule | Name: Cytoplasmic dynein 1 intermediate chain 2 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 68.442141 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MSDKSELKAE LERKKQRLAQ IREEKKRKEE ERKKKETDQK KEAVAPVQEE SDLEKKRREA EALLQSMGLT PESPIVPPPM SPSSKSVST PSEAGSQDSG DGAVGSRRGP IKLGMAKITQ VDFPPREIVT YTKETQTPVM AQPKEDEEED DDVVAPKPPI E PEEEKTLK ...String: MSDKSELKAE LERKKQRLAQ IREEKKRKEE ERKKKETDQK KEAVAPVQEE SDLEKKRREA EALLQSMGLT PESPIVPPPM SPSSKSVST PSEAGSQDSG DGAVGSRRGP IKLGMAKITQ VDFPPREIVT YTKETQTPVM AQPKEDEEED DDVVAPKPPI E PEEEKTLK KDEENDSKAP PHELTEEEKQ QILHSEEFLS FFDHSTRIVE RALSEQINIF FDYSGRDLED KEGEIQAGAK LS LNRQFFD ERWSKHRVVS CLDWSSQYPE LLVASYNNNE DAPHEPDGVA LVWNMKYKKT TPEYVFHCQS AVMSATFAKF HPN LVVGGT YSGQIVLWDN RSNKRTPVQR TPLSAAAHTH PVYCVNVVGT QNAHNLISIS TDGKICSWSL DMLSHPQDSM ELVH KQSKA VAVTSMSFPV GDVNNFVVGS EEGSVYTACR HGSKAGISEM FEGHQGPITG IHCHAAVGAV DFSHLFVTSS FDWTV KLWS TKNNKPLYSF EDNAGYVYDV MWSPTHPALF ACVDGMGRLD LWNLNNDTEV PTASISVEGN PALNRVRWTH SGREIA VGD SEGQIVIYDV GEQIAVPRND EWARFGRTLA EINANRADAE EEAATRIPA UniProtKB: Cytoplasmic dynein 1 intermediate chain 2 |
-Macromolecule #4: Cytoplasmic dynein 1 light intermediate chain 2
| Macromolecule | Name: Cytoplasmic dynein 1 light intermediate chain 2 / type: protein_or_peptide / ID: 4 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 54.173156 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MAPVGVEKKL LLGPNGPAVA AAGDLTSEEE EGQSLWSSIL SEVSTRARSK LPSGKNILVF GEDGSGKTTL MTKLQGAEHG KKGRGLEYL YLSVHDEDRD DHTRCNVWIL DGDLYHKGLL KFAVSAESLP ETLVIFVADM SRPWTVMESL QKWASVLREH I DKMKIPPE ...String: MAPVGVEKKL LLGPNGPAVA AAGDLTSEEE EGQSLWSSIL SEVSTRARSK LPSGKNILVF GEDGSGKTTL MTKLQGAEHG KKGRGLEYL YLSVHDEDRD DHTRCNVWIL DGDLYHKGLL KFAVSAESLP ETLVIFVADM SRPWTVMESL QKWASVLREH I DKMKIPPE KMRELERKFV KDFQDYMEPE EGCQGSPQRR GPLTSGSDEE NVALPLGDNV LTHNLGIPVL VVCTKCDAVS VL EKEHDYR DEHLDFIQSH LRRFCLQYGA ALIYTSVKEE KNLDLLYKYI VHKTYGFHFT TPALVVEKDA VFIPAGWDNE KKI AILHEN FTTVKPEDAY EDFIVKPPVR KLVHDKELAA EDEQVFLMKQ QSLLAKQPAT PTRASESPAR GPSGSPRTQG RGGP ASVPS SSPGTSVKKP DPNIKNNAAS EGVLASFFNS LLSKKTGSPG SPGAGGVQST AKKSGQKTVL SNVQEELDRM TRKPD SMVT NSSTENEA UniProtKB: Cytoplasmic dynein 1 light intermediate chain 2 |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.2 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 4.0 µm / Nominal defocus min: 0.5 µm Bright-field microscopy / Nominal defocus max: 4.0 µm / Nominal defocus min: 0.5 µm |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Average electron dose: 53.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
 Movie
Movie Controller
Controller


































 Z
Z Y
Y X
X