[English] 日本語
 Yorodumi
Yorodumi- EMDB-40617: Cryo-EM structure of the human nucleosome core particle in comple... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
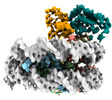
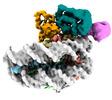
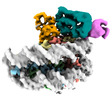
| Title | Cryo-EM structure of the human nucleosome core particle in complex with RNF168 and UbcH5c with backside ubiquitin (UbcH5c chemically conjugated to histone H2A) (class 1) | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords |  Nucleosome core particle / Nucleosome core particle /  chromatin / RNF168 / chromatin / RNF168 /  RING domain / UbcH5c / RING domain / UbcH5c /  DNA repair / DNA double-strand break / DNA repair / DNA double-strand break /  Homologous recombination / Homologous recombination /  53BP1 / 53BP1 /  ubiquitin / ubiquitin /  STRUCTURAL PROTEIN-DNA-TRANSFERASE complex / STRUCTURAL PROTEIN-DNA-TRANSFERASE complex /  TRANSFERASE / TRANSFERASE-DNA complex TRANSFERASE / TRANSFERASE-DNA complex | |||||||||
| Function / homology |  Function and homology information Function and homology informationhistone H2AK15 ubiquitin ligase activity / histone ubiquitin ligase activity / (E3-independent) E2 ubiquitin-conjugating enzyme / protein K6-linked ubiquitination / Signaling by BMP / double-strand break repair via classical nonhomologous end joining /  isotype switching / protein K11-linked ubiquitination / hypothalamus gonadotrophin-releasing hormone neuron development / DNA repair-dependent chromatin remodeling ...histone H2AK15 ubiquitin ligase activity / histone ubiquitin ligase activity / (E3-independent) E2 ubiquitin-conjugating enzyme / protein K6-linked ubiquitination / Signaling by BMP / double-strand break repair via classical nonhomologous end joining / isotype switching / protein K11-linked ubiquitination / hypothalamus gonadotrophin-releasing hormone neuron development / DNA repair-dependent chromatin remodeling ...histone H2AK15 ubiquitin ligase activity / histone ubiquitin ligase activity / (E3-independent) E2 ubiquitin-conjugating enzyme / protein K6-linked ubiquitination / Signaling by BMP / double-strand break repair via classical nonhomologous end joining /  isotype switching / protein K11-linked ubiquitination / hypothalamus gonadotrophin-releasing hormone neuron development / DNA repair-dependent chromatin remodeling / female meiosis I / positive regulation of protein monoubiquitination / mitochondrion transport along microtubule / positive regulation of protein targeting to mitochondrion / isotype switching / protein K11-linked ubiquitination / hypothalamus gonadotrophin-releasing hormone neuron development / DNA repair-dependent chromatin remodeling / female meiosis I / positive regulation of protein monoubiquitination / mitochondrion transport along microtubule / positive regulation of protein targeting to mitochondrion /  E2 ubiquitin-conjugating enzyme / fat pad development / K63-linked polyubiquitin modification-dependent protein binding / response to ionizing radiation / female gonad development / seminiferous tubule development / male meiosis I / negative regulation of transcription elongation by RNA polymerase II / ubiquitin conjugating enzyme activity / protein monoubiquitination / positive regulation of intrinsic apoptotic signaling pathway by p53 class mediator / protein K63-linked ubiquitination / negative regulation of BMP signaling pathway / E2 ubiquitin-conjugating enzyme / fat pad development / K63-linked polyubiquitin modification-dependent protein binding / response to ionizing radiation / female gonad development / seminiferous tubule development / male meiosis I / negative regulation of transcription elongation by RNA polymerase II / ubiquitin conjugating enzyme activity / protein monoubiquitination / positive regulation of intrinsic apoptotic signaling pathway by p53 class mediator / protein K63-linked ubiquitination / negative regulation of BMP signaling pathway /  nucleosome binding / negative regulation of tumor necrosis factor-mediated signaling pathway / protein K48-linked ubiquitination / negative regulation of megakaryocyte differentiation / protein autoubiquitination / protein localization to CENP-A containing chromatin / Chromatin modifying enzymes / Replacement of protamines by nucleosomes in the male pronucleus / interstrand cross-link repair / CENP-A containing nucleosome / nucleosome binding / negative regulation of tumor necrosis factor-mediated signaling pathway / protein K48-linked ubiquitination / negative regulation of megakaryocyte differentiation / protein autoubiquitination / protein localization to CENP-A containing chromatin / Chromatin modifying enzymes / Replacement of protamines by nucleosomes in the male pronucleus / interstrand cross-link repair / CENP-A containing nucleosome /  ubiquitin ligase complex / SUMOylation of DNA damage response and repair proteins / epigenetic regulation of gene expression / ubiquitin ligase complex / SUMOylation of DNA damage response and repair proteins / epigenetic regulation of gene expression /  energy homeostasis / regulation of neuron apoptotic process / Packaging Of Telomere Ends / regulation of proteasomal protein catabolic process / Recognition and association of DNA glycosylase with site containing an affected purine / Cleavage of the damaged purine / Maturation of protein E / Maturation of protein E / ER Quality Control Compartment (ERQC) / Deposition of new CENPA-containing nucleosomes at the centromere / Myoclonic epilepsy of Lafora / FLT3 signaling by CBL mutants / Prevention of phagosomal-lysosomal fusion / IRAK2 mediated activation of TAK1 complex / Alpha-protein kinase 1 signaling pathway / energy homeostasis / regulation of neuron apoptotic process / Packaging Of Telomere Ends / regulation of proteasomal protein catabolic process / Recognition and association of DNA glycosylase with site containing an affected purine / Cleavage of the damaged purine / Maturation of protein E / Maturation of protein E / ER Quality Control Compartment (ERQC) / Deposition of new CENPA-containing nucleosomes at the centromere / Myoclonic epilepsy of Lafora / FLT3 signaling by CBL mutants / Prevention of phagosomal-lysosomal fusion / IRAK2 mediated activation of TAK1 complex / Alpha-protein kinase 1 signaling pathway /  Glycogen synthesis / IRAK1 recruits IKK complex / IRAK1 recruits IKK complex upon TLR7/8 or 9 stimulation / Membrane binding and targetting of GAG proteins / Endosomal Sorting Complex Required For Transport (ESCRT) / IRAK2 mediated activation of TAK1 complex upon TLR7/8 or 9 stimulation / PTK6 Regulates RTKs and Their Effectors AKT1 and DOK1 / Negative regulation of FLT3 / Recognition and association of DNA glycosylase with site containing an affected pyrimidine / Cleavage of the damaged pyrimidine / Constitutive Signaling by NOTCH1 HD Domain Mutants / Regulation of FZD by ubiquitination / TICAM1,TRAF6-dependent induction of TAK1 complex / NOTCH2 Activation and Transmission of Signal to the Nucleus / TICAM1-dependent activation of IRF3/IRF7 / APC/C:Cdc20 mediated degradation of Cyclin B / Inhibition of DNA recombination at telomere / Meiotic synapsis / p75NTR recruits signalling complexes / Downregulation of ERBB4 signaling / positive regulation of DNA repair / TRAF6 mediated IRF7 activation in TLR7/8 or 9 signaling / APC-Cdc20 mediated degradation of Nek2A / PINK1-PRKN Mediated Mitophagy / TRAF6-mediated induction of TAK1 complex within TLR4 complex / telomere organization / Pexophagy / Regulation of innate immune responses to cytosolic DNA / InlA-mediated entry of Listeria monocytogenes into host cells / VLDLR internalisation and degradation / Downregulation of ERBB2:ERBB3 signaling / RNA Polymerase I Promoter Opening / NF-kB is activated and signals survival / NRIF signals cell death from the nucleus / Interleukin-7 signaling / Regulation of PTEN localization / Activated NOTCH1 Transmits Signal to the Nucleus / Glycogen synthesis / IRAK1 recruits IKK complex / IRAK1 recruits IKK complex upon TLR7/8 or 9 stimulation / Membrane binding and targetting of GAG proteins / Endosomal Sorting Complex Required For Transport (ESCRT) / IRAK2 mediated activation of TAK1 complex upon TLR7/8 or 9 stimulation / PTK6 Regulates RTKs and Their Effectors AKT1 and DOK1 / Negative regulation of FLT3 / Recognition and association of DNA glycosylase with site containing an affected pyrimidine / Cleavage of the damaged pyrimidine / Constitutive Signaling by NOTCH1 HD Domain Mutants / Regulation of FZD by ubiquitination / TICAM1,TRAF6-dependent induction of TAK1 complex / NOTCH2 Activation and Transmission of Signal to the Nucleus / TICAM1-dependent activation of IRF3/IRF7 / APC/C:Cdc20 mediated degradation of Cyclin B / Inhibition of DNA recombination at telomere / Meiotic synapsis / p75NTR recruits signalling complexes / Downregulation of ERBB4 signaling / positive regulation of DNA repair / TRAF6 mediated IRF7 activation in TLR7/8 or 9 signaling / APC-Cdc20 mediated degradation of Nek2A / PINK1-PRKN Mediated Mitophagy / TRAF6-mediated induction of TAK1 complex within TLR4 complex / telomere organization / Pexophagy / Regulation of innate immune responses to cytosolic DNA / InlA-mediated entry of Listeria monocytogenes into host cells / VLDLR internalisation and degradation / Downregulation of ERBB2:ERBB3 signaling / RNA Polymerase I Promoter Opening / NF-kB is activated and signals survival / NRIF signals cell death from the nucleus / Interleukin-7 signaling / Regulation of PTEN localization / Activated NOTCH1 Transmits Signal to the Nucleus /  Regulation of BACH1 activity / Assembly of the ORC complex at the origin of replication / Translesion synthesis by REV1 / SUMOylation of chromatin organization proteins / Synthesis of active ubiquitin: roles of E1 and E2 enzymes / Translesion synthesis by POLK / MAP3K8 (TPL2)-dependent MAPK1/3 activation / TICAM1, RIP1-mediated IKK complex recruitment Regulation of BACH1 activity / Assembly of the ORC complex at the origin of replication / Translesion synthesis by REV1 / SUMOylation of chromatin organization proteins / Synthesis of active ubiquitin: roles of E1 and E2 enzymes / Translesion synthesis by POLK / MAP3K8 (TPL2)-dependent MAPK1/3 activation / TICAM1, RIP1-mediated IKK complex recruitmentSimilarity search - Function | |||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.9 Å cryo EM / Resolution: 3.9 Å | |||||||||
 Authors Authors | Hu Q / Botuyan MV / Zhao D / Cui G / Mer G | |||||||||
| Funding support |  United States, 2 items United States, 2 items
| |||||||||
 Citation Citation |  Journal: Mol Cell / Year: 2024 Journal: Mol Cell / Year: 2024Title: Mechanisms of RNF168 nucleosome recognition and ubiquitylation. Authors: Qi Hu / Debiao Zhao / Gaofeng Cui / Janarjan Bhandari / James R Thompson / Maria Victoria Botuyan / Georges Mer /  Abstract: RNF168 plays a central role in the DNA damage response (DDR) by ubiquitylating histone H2A at K13 and K15. These modifications direct BRCA1-BARD1 and 53BP1 foci formation in chromatin, essential for ...RNF168 plays a central role in the DNA damage response (DDR) by ubiquitylating histone H2A at K13 and K15. These modifications direct BRCA1-BARD1 and 53BP1 foci formation in chromatin, essential for cell-cycle-dependent DNA double-strand break (DSB) repair pathway selection. The mechanism by which RNF168 catalyzes the targeted accumulation of H2A ubiquitin conjugates to form repair foci around DSBs remains unclear. Here, using cryoelectron microscopy (cryo-EM), nuclear magnetic resonance (NMR) spectroscopy, and functional assays, we provide a molecular description of the reaction cycle and dynamics of RNF168 as it modifies the nucleosome and recognizes its ubiquitylation products. We demonstrate an interaction of a canonical ubiquitin-binding domain within full-length RNF168, which not only engages ubiquitin but also the nucleosome surface, clarifying how such site-specific ubiquitin recognition propels a signal amplification loop. Beyond offering mechanistic insights into a key DDR protein, our study aids in understanding site specificity in both generating and interpreting chromatin ubiquitylation. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_40617.map.gz emd_40617.map.gz | 38.2 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-40617-v30.xml emd-40617-v30.xml emd-40617.xml emd-40617.xml | 28 KB 28 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_40617.png emd_40617.png | 97.3 KB | ||
| Masks |  emd_40617_msk_1.map emd_40617_msk_1.map | 64 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-40617.cif.gz emd-40617.cif.gz | 7.7 KB | ||
| Others |  emd_40617_half_map_1.map.gz emd_40617_half_map_1.map.gz emd_40617_half_map_2.map.gz emd_40617_half_map_2.map.gz | 50 MB 50 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-40617 http://ftp.pdbj.org/pub/emdb/structures/EMD-40617 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40617 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-40617 | HTTPS FTP |
-Related structure data
| Related structure data |  8sn9MC  8smwC  8smxC  8smyC  8smzC  8sn0C  8sn1C  8sn2C  8sn3C  8sn4C  8sn5C  8sn6C  8sn7C  8sn8C  8snaC  8txvC  8txwC  8txxC  8u13C  8u14C  8upfC  8uq8C  8uq9C  8uqaC  8uqbC  8uqcC  8uqdC  8uqeC M: atomic model generated by this map C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_40617.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_40617.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.328 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_40617_msk_1.map emd_40617_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #1
| File | emd_40617_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: #2
| File | emd_40617_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : Human nucleosome core particle in complex with RNF168 and UbcH5c~...
+Supramolecule #1: Human nucleosome core particle in complex with RNF168 and UbcH5c~...
+Macromolecule #1: Histone H3.1
+Macromolecule #2: Histone H4
+Macromolecule #3: Histone H2A type 1-B/E
+Macromolecule #4: Histone H2B type 1-J
+Macromolecule #7: E3 ubiquitin-protein ligase RNF168
+Macromolecule #8: Ubiquitin-conjugating enzyme E2 D3
+Macromolecule #9: Polyubiquitin-B
+Macromolecule #5: DNA (147-MER)
+Macromolecule #6: DNA (147-MER)
+Macromolecule #10: ZINC ION
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 0.4 mg/mL |
|---|---|
| Buffer | pH: 7.5 |
| Grid | Model: Quantifoil R1.2/1.3 / Material: COPPER / Pretreatment - Type: GLOW DISCHARGE / Pretreatment - Time: 60 sec. |
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277 K / Instrument: FEI VITROBOT MARK IV |
| Details | 10 mM HEPES, 100 mM NaCl, 1 mM DTT, pH 7.5 |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 130000 Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 3.0 µm / Nominal defocus min: 0.5 µm / Nominal magnification: 130000 |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Number grids imaged: 1 / Number real images: 14179 / Average electron dose: 60.0 e/Å2 Details: 14179 images were recorded in movie-mode of which 14113 were retained for particle picking. |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Particle selection | Number selected: 16063830 |
|---|---|
| Startup model | Type of model: NONE |
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC (ver. 4.12) |
| Final 3D classification | Software - Name: cryoSPARC (ver. 4.12) |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC (ver. 4.12) |
| Final reconstruction | Applied symmetry - Point group: C1 (asymmetric) / Algorithm: FOURIER SPACE / Resolution.type: BY AUTHOR / Resolution: 3.9 Å / Resolution method: FSC 0.143 CUT-OFF / Software - Name: RELION (ver. 3.0) / Number images used: 33118 |
-Atomic model buiding 1
| Initial model |
| ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Space: REAL / Protocol: RIGID BODY FIT | ||||||||||
| Output model |  PDB-8sn9: |
 Movie
Movie Controller
Controller













































 Z
Z Y
Y X
X






























