[English] 日本語
 Yorodumi
Yorodumi- EMDB-33924: Cryo-EM structure of Nb29-alpha1AAR-miniGsq complex bound to oxym... -
+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM structure of Nb29-alpha1AAR-miniGsq complex bound to oxymetazoline | |||||||||
 Map data Map data | ||||||||||
 Sample Sample |
| |||||||||
 Keywords Keywords |  GPCR / GPCR /  Nanobody / Nanobody /  Agonist / Agonist /  Complex / Complex /  MEMBRANE PROTEIN MEMBRANE PROTEIN | |||||||||
| Biological species | synthetic construct (others) /   Homo sapiens (human) Homo sapiens (human) | |||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 2.92 Å cryo EM / Resolution: 2.92 Å | |||||||||
 Authors Authors | Toyoda Y / Zhu A / Yan C / Kobilka BK / Liu X | |||||||||
| Funding support |  China, 1 items China, 1 items
| |||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Structural basis of α-adrenergic receptor activation and recognition by an extracellular nanobody. Authors: Yosuke Toyoda / Angqi Zhu / Fang Kong / Sisi Shan / Jiawei Zhao / Nan Wang / Xiaoou Sun / Linqi Zhang / Chuangye Yan / Brian K Kobilka / Xiangyu Liu /    Abstract: The αadrenergic receptor (αAR) belongs to the family of G protein-coupled receptors that respond to adrenaline and noradrenaline. αAR is involved in smooth muscle contraction and cognitive ...The αadrenergic receptor (αAR) belongs to the family of G protein-coupled receptors that respond to adrenaline and noradrenaline. αAR is involved in smooth muscle contraction and cognitive function. Here, we present three cryo-electron microscopy structures of human αAR bound to the endogenous agonist noradrenaline, its selective agonist oxymetazoline, and the antagonist tamsulosin, with resolutions range from 2.9 Å to 3.5 Å. Our active and inactive αAR structures reveal the activation mechanism and distinct ligand binding modes for noradrenaline compared with other adrenergic receptor subtypes. In addition, we identified a nanobody that preferentially binds to the extracellular vestibule of αAR when bound to the selective agonist oxymetazoline. These results should facilitate the design of more selective therapeutic drugs targeting both orthosteric and allosteric sites in this receptor family. | |||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_33924.map.gz emd_33924.map.gz | 59.7 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-33924-v30.xml emd-33924-v30.xml emd-33924.xml emd-33924.xml | 18.4 KB 18.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_33924.png emd_33924.png | 66.1 KB | ||
| Others |  emd_33924_half_map_1.map.gz emd_33924_half_map_1.map.gz emd_33924_half_map_2.map.gz emd_33924_half_map_2.map.gz | 59.5 MB 59.5 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-33924 http://ftp.pdbj.org/pub/emdb/structures/EMD-33924 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33924 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-33924 | HTTPS FTP |
-Related structure data
| Related structure data |  7ym8MC  7ymhC  7ymjC M: atomic model generated by this map C: citing same article ( |
|---|
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|
- Map
Map
| File |  Download / File: emd_33924.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_33924.map.gz / Format: CCP4 / Size: 64 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.0825 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Half map: #2
| File | emd_33924_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
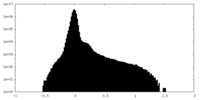
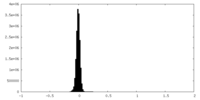
| Density Histograms |
-Half map: #1
| File | emd_33924_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
-Entire : Nb29-alpha1AAR-miniGsq complex
| Entire | Name: Nb29-alpha1AAR-miniGsq complex |
|---|---|
| Components |
|
-Supramolecule #1: Nb29-alpha1AAR-miniGsq complex
| Supramolecule | Name: Nb29-alpha1AAR-miniGsq complex / type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#3 |
|---|
-Supramolecule #2: Nb29
| Supramolecule | Name: Nb29 / type: complex / ID: 2 / Parent: 1 / Macromolecule list: #3 |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
-Supramolecule #3: alpha1AAR
| Supramolecule | Name: alpha1AAR / type: complex / ID: 3 / Parent: 1 / Macromolecule list: #2 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
-Supramolecule #4: miniGsq
| Supramolecule | Name: miniGsq / type: complex / ID: 4 / Parent: 1 / Macromolecule list: #1 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
-Macromolecule #1: miniGsq
| Macromolecule | Name: miniGsq / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 28.571365 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MGHHHHHHHH LEVLFQGPIE KQLQKDKQVY RATHRLLLLG ADNSGKSTIV KQMRILHGGS GGSGGTSGIF ETKFQVDKVN FHMFDVGGQ RDERRKWIQC FNDVTAIIFV VDSSDYNRLQ EALNDFKSIW NNRWLRTISV ILFLNKQDLL AEKVLAGKSK I EDYFPEFA ...String: MGHHHHHHHH LEVLFQGPIE KQLQKDKQVY RATHRLLLLG ADNSGKSTIV KQMRILHGGS GGSGGTSGIF ETKFQVDKVN FHMFDVGGQ RDERRKWIQC FNDVTAIIFV VDSSDYNRLQ EALNDFKSIW NNRWLRTISV ILFLNKQDLL AEKVLAGKSK I EDYFPEFA RYTTPEDATP EPGEDPRVTR AKYFIRDEFL RISTASGDGR HYCYPHFTCA VDTENARRIF NDCKDIILQM NL REYNLV |
-Macromolecule #2: alpha1A adrenergic receptor
| Macromolecule | Name: alpha1A adrenergic receptor / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 53.736715 KDa |
| Recombinant expression | Organism:   Spodoptera frugiperda (fall armyworm) Spodoptera frugiperda (fall armyworm) |
| Sequence | String: MKTIIALSYI FCLVFADYKD DDDAMVFLSG QASDSSQCTQ PPAPVQISKA ILLGVILGGL ILFGVLGNIL VILSVACHRH LHSVTHYYI VNLAVADLLL TSTVLPFSAI FEVLGYWAFG RVFCNIWAAV DVLCCTASIM GLCIISIDRY IGVSYPLRYP T IVTQRRGL ...String: MKTIIALSYI FCLVFADYKD DDDAMVFLSG QASDSSQCTQ PPAPVQISKA ILLGVILGGL ILFGVLGNIL VILSVACHRH LHSVTHYYI VNLAVADLLL TSTVLPFSAI FEVLGYWAFG RVFCNIWAAV DVLCCTASIM GLCIISIDRY IGVSYPLRYP T IVTQRRGL MALLCVWALS LVISIGPLFG WRQPAPEDET ICQINEEPGY VLFSALGSFY LPLAIILVMY CRVYVVAKRE SR GLKSGLN IFEMLRIDEG GGSGGDEAEK LFNQDVDAAV RGILRNAKLK PVYDSLDAVR RAALINMVFQ MGETGVAGFT NSL RMLQQK RWDEAAVNLA KSRWYNQTPN RAKRVITTFR TGTWDAYLKF SREKKAAKTL GIVVGCFVLC WLPFFLVMPI GSFF PDFKP SETVFKIVFW LGYLNSCINP IIYPCSSQEF KKAFQNVLRI QCLCRKQSSK HALGYTLHPP SQAVEGQHHH HHHHH |
-Macromolecule #3: nanobody 29
| Macromolecule | Name: nanobody 29 / type: protein_or_peptide / ID: 3 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism: synthetic construct (others) |
| Molecular weight | Theoretical: 15.815713 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MKYLLPTAAA GLLLLAAQPA MAQVQLQESG GGLVQAGGSL RLSCAASGNI SAHWKMGWYR QAPGKEREFV AGIGYANTNY ADSVKGRFT ISRDNAKNTV YLQMNSLKPE DTAVYYCAAY SYYRDHSYWG QGTQVTVSSH HHHHH |
-Macromolecule #4: Oxymetazoline
| Macromolecule | Name: Oxymetazoline / type: ligand / ID: 4 / Number of copies: 1 / Formula: J5C |
|---|---|
| Molecular weight | Theoretical: 260.375 Da |
| Chemical component information |  ChemComp-J5C: |
-Macromolecule #5: CHOLESTEROL HEMISUCCINATE
| Macromolecule | Name: CHOLESTEROL HEMISUCCINATE / type: ligand / ID: 5 / Number of copies: 1 / Formula: Y01 |
|---|---|
| Molecular weight | Theoretical: 486.726 Da |
| Chemical component information | 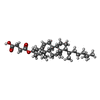 ChemComp-Y01: |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Nominal defocus max: 1.8 µm / Nominal defocus min: 1.3 µm Bright-field microscopy / Nominal defocus max: 1.8 µm / Nominal defocus min: 1.3 µm |
| Image recording | Film or detector model: GATAN K3 (6k x 4k) / Average electron dose: 50.0 e/Å2 |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: OTHER |
|---|---|
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: cryoSPARC |
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 2.92 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 359000 |
 Movie
Movie Controller
Controller



 Z
Z Y
Y X
X