+ Open data
Open data
- Basic information
Basic information
| Entry | Database: EMDB / ID: EMD-11339 | |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Kinesin binding protein complexed with Kif15 motor domain | |||||||||||||||||||||
 Map data Map data | Kinesin binding protein (KBP) complexed with Kif15 motor domain sharpened/filtered cryo-EM density. | |||||||||||||||||||||
 Sample Sample |
| |||||||||||||||||||||
 Keywords Keywords |  Kinesin / Kinesin /  microtubules / kinesin binding protein / KBP / microtubules / kinesin binding protein / KBP /  MOTOR PROTEIN MOTOR PROTEIN | |||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology informationplus-end kinesin complex /  central nervous system projection neuron axonogenesis / Kinesins / COPI-dependent Golgi-to-ER retrograde traffic / central nervous system projection neuron axonogenesis / Kinesins / COPI-dependent Golgi-to-ER retrograde traffic /  kinesin complex / kinesin complex /  microtubule motor activity / mitochondrial transport / microtubule-based movement / cytoskeletal motor activity / microtubule motor activity / mitochondrial transport / microtubule-based movement / cytoskeletal motor activity /  kinesin binding ...plus-end kinesin complex / kinesin binding ...plus-end kinesin complex /  central nervous system projection neuron axonogenesis / Kinesins / COPI-dependent Golgi-to-ER retrograde traffic / central nervous system projection neuron axonogenesis / Kinesins / COPI-dependent Golgi-to-ER retrograde traffic /  kinesin complex / kinesin complex /  microtubule motor activity / mitochondrial transport / microtubule-based movement / cytoskeletal motor activity / microtubule motor activity / mitochondrial transport / microtubule-based movement / cytoskeletal motor activity /  kinesin binding / MHC class II antigen presentation / neuron projection maintenance / spindle / microtubule cytoskeleton organization / mitotic cell cycle / kinesin binding / MHC class II antigen presentation / neuron projection maintenance / spindle / microtubule cytoskeleton organization / mitotic cell cycle /  microtubule binding / microtubule binding /  microtubule / in utero embryonic development / microtubule / in utero embryonic development /  cytoskeleton / cytoskeleton /  centrosome / centrosome /  ATP hydrolysis activity / ATP hydrolysis activity /  mitochondrion / mitochondrion /  ATP binding / ATP binding /  membrane / membrane /  cytosol cytosolSimilarity search - Function | |||||||||||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human) | |||||||||||||||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 6.9 Å cryo EM / Resolution: 6.9 Å | |||||||||||||||||||||
 Authors Authors | Atherton J / Hummel JJA | |||||||||||||||||||||
| Funding support |  United Kingdom, United Kingdom,  Switzerland, Switzerland,  United States, 6 items United States, 6 items
| |||||||||||||||||||||
 Citation Citation |  Journal: Elife / Year: 2020 Journal: Elife / Year: 2020Title: The mechanism of kinesin inhibition by kinesin-binding protein. Authors: Joseph Atherton / Jessica Ja Hummel / Natacha Olieric / Julia Locke / Alejandro Peña / Steven S Rosenfeld / Michel O Steinmetz / Casper C Hoogenraad / Carolyn A Moores /     Abstract: Subcellular compartmentalisation is necessary for eukaryotic cell function. Spatial and temporal regulation of kinesin activity is essential for building these local environments via control of ...Subcellular compartmentalisation is necessary for eukaryotic cell function. Spatial and temporal regulation of kinesin activity is essential for building these local environments via control of intracellular cargo distribution. Kinesin-binding protein (KBP) interacts with a subset of kinesins via their motor domains, inhibits their microtubule (MT) attachment, and blocks their cellular function. However, its mechanisms of inhibition and selectivity have been unclear. Here we use cryo-electron microscopy to reveal the structure of KBP and of a KBP-kinesin motor domain complex. KBP is a tetratricopeptide repeat-containing, right-handed α-solenoid that sequesters the kinesin motor domain's tubulin-binding surface, structurally distorting the motor domain and sterically blocking its MT attachment. KBP uses its α-solenoid concave face and edge loops to bind the kinesin motor domain, and selected structure-guided mutations disrupt KBP inhibition of kinesin transport in cells. The KBP-interacting motor domain surface contains motifs exclusively conserved in KBP-interacting kinesins, suggesting a basis for kinesin selectivity. | |||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Movie |
 Movie viewer Movie viewer |
|---|---|
| Structure viewer | EM map:  SurfView SurfView Molmil Molmil Jmol/JSmol Jmol/JSmol |
| Supplemental images |
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_11339.map.gz emd_11339.map.gz | 14.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-11339-v30.xml emd-11339-v30.xml emd-11339.xml emd-11339.xml | 13.5 KB 13.5 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_11339.png emd_11339.png | 127.2 KB | ||
| Filedesc metadata |  emd-11339.cif.gz emd-11339.cif.gz | 6 KB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-11339 http://ftp.pdbj.org/pub/emdb/structures/EMD-11339 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11339 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-11339 | HTTPS FTP |
-Related structure data
| Related structure data |  6zphMC  6zpgC  6zpiC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_11339.map.gz / Format: CCP4 / Size: 22.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_11339.map.gz / Format: CCP4 / Size: 22.2 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Kinesin binding protein (KBP) complexed with Kif15 motor domain sharpened/filtered cryo-EM density. | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Voxel size | X=Y=Z: 1.048 Å | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Density |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Details | EMDB XML:
CCP4 map header:
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
-Supplemental data
- Sample components
Sample components
-Entire : Kinesin binding protein complexed with Kif15 motor domain cryo-EM...
| Entire | Name: Kinesin binding protein complexed with Kif15 motor domain cryo-EM density |
|---|---|
| Components |
|
-Supramolecule #1: Kinesin binding protein complexed with Kif15 motor domain cryo-EM...
| Supramolecule | Name: Kinesin binding protein complexed with Kif15 motor domain cryo-EM density type: complex / ID: 1 / Parent: 0 / Macromolecule list: #1-#2 |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 72 KDa |
-Macromolecule #1: KIF-binding protein
| Macromolecule | Name: KIF-binding protein / type: protein_or_peptide / ID: 1 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 71.913945 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MANVPWAEVC EKFQAALALS RVELHKNPEK EPYKSKYSAR ALLEEVKALL GPAPEDEDER PEAEDGPGAG DHALGLPAEV VEPEGPVAQ RAVRLAVIEF HLGVNHIDTE ELSAGEEHLV KCLRLLRRYR LSHDCISLCI QAQNNLGILW SEREEIETAQ A YLESSEAL ...String: MANVPWAEVC EKFQAALALS RVELHKNPEK EPYKSKYSAR ALLEEVKALL GPAPEDEDER PEAEDGPGAG DHALGLPAEV VEPEGPVAQ RAVRLAVIEF HLGVNHIDTE ELSAGEEHLV KCLRLLRRYR LSHDCISLCI QAQNNLGILW SEREEIETAQ A YLESSEAL YNQYMKEVGS PPLDPTERFL PEEEKLTEQE RSKRFEKVYT HNLYYLAQVY QHLEMFEKAA HYCHSTLKRQ LE HNAYHPI EWAINAATLS QFYINKLCFM EARHCLSAAN VIFGQTGKIS ATEDTPEAEG EVPELYHQRK GEIARCWIKY CLT LMQNAQ LSMQDNIGEL DLDKQSELRA LRKKELDEEE SIRKKAVQFG TGELCDAISA VEEKVSYLRP LDFEEARELF LLGQ HYVFE AKEFFQIDGY VTDHIEVVQD HSALFKVLAF FETDMERRCK MHKRRIAMLE PLTVDLNPQY YLLVNRQIQF EIAHA YYDM MDLKVAIADR LRDPDSHIVK KINNLNKSAL KYYQLFLDSL RDPNKVFPEH IGEDVLRPAM LAKFRVARLY GKIITA DPK KELENLATSL EHYKFIVDYC EKHPEAAQEI EVELELSKEM VSLLPTKMER FRTKMALT UniProtKB: KIF-binding protein |
-Macromolecule #2: Kinesin-like protein KIF15
| Macromolecule | Name: Kinesin-like protein KIF15 / type: protein_or_peptide / ID: 2 / Number of copies: 1 / Enantiomer: LEVO |
|---|---|
| Source (natural) | Organism:   Homo sapiens (human) Homo sapiens (human) |
| Molecular weight | Theoretical: 41.986004 KDa |
| Recombinant expression | Organism:   Escherichia coli (E. coli) Escherichia coli (E. coli) |
| Sequence | String: MAPGSKTELR SVTNGQSNQP SNEGDAIKVF VRIRPPAERS GSADGEQNLS LSVLSSTSLR LHSNPEPKTF TFDHVADVDT TQESVFATV AKSIVESCMS GYNGTIFAYG QTGSGKTFTM MGPSESDNFS HNLRGVIPRS FEYLFSLIDR EKEKAGAGKS F LSKCSFIE ...String: MAPGSKTELR SVTNGQSNQP SNEGDAIKVF VRIRPPAERS GSADGEQNLS LSVLSSTSLR LHSNPEPKTF TFDHVADVDT TQESVFATV AKSIVESCMS GYNGTIFAYG QTGSGKTFTM MGPSESDNFS HNLRGVIPRS FEYLFSLIDR EKEKAGAGKS F LSKCSFIE IYNEQIYDLL DSASAGLYLR EHIKKGVFVV GAVEQVVTSA AEAYQVLSGG WRNRRVASTS MNRESSRSHA VF TITIESM EKCNEIVNIR TSLLNLVDLA GSERQKDTHA EGMRLKEAGN INRSLSTLGQ VITALVDVGN GKQRHVSYRD SKL TFLLRD SLGGNAKTAI IANVHPGSRS FGETLSTLNF AQRAKLIKNK AVVNEDTQCL EHHHHHH UniProtKB: Kinesin-like protein KIF15 |
-Macromolecule #3: ADENOSINE-5'-DIPHOSPHATE
| Macromolecule | Name: ADENOSINE-5'-DIPHOSPHATE / type: ligand / ID: 3 / Number of copies: 1 / Formula: ADP |
|---|---|
| Molecular weight | Theoretical: 427.201 Da |
| Chemical component information | 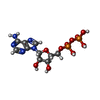 ChemComp-ADP: |
-Macromolecule #4: MAGNESIUM ION
| Macromolecule | Name: MAGNESIUM ION / type: ligand / ID: 4 / Number of copies: 1 / Formula: MG |
|---|---|
| Molecular weight | Theoretical: 24.305 Da |
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Buffer | pH: 7.5 |
|---|---|
| Vitrification | Cryogen name: ETHANE |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy Bright-field microscopy |
| Image recording | Film or detector model: GATAN K2 SUMMIT (4k x 4k) / Detector mode: COUNTING / Average electron dose: 42.0 e/Å2 / Details: Movies were dose weighted. |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: INSILICO MODEL / Details: De novo generated model in cryoSPARC2. |
|---|---|
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD / Software - Name: RELION |
| Final reconstruction | Resolution.type: BY AUTHOR / Resolution: 6.9 Å / Resolution method: FSC 0.143 CUT-OFF / Number images used: 7513 |
 Movie
Movie Controller
Controller





























