+ Open data
Open data
- Basic information
Basic information
| Entry |  | |||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
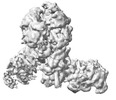
| Title | Yeast replisome in state I | |||||||||||||||||||||||||||
 Map data Map data | ||||||||||||||||||||||||||||
 Sample Sample |
| |||||||||||||||||||||||||||
 Keywords Keywords |  replisome / replisome /  complex / complex /  DNA replication / DNA replication /  REPLICATION REPLICATION | |||||||||||||||||||||||||||
| Function / homology |  Function and homology information Function and homology information establishment of sister chromatid cohesion / maintenance of DNA repeat elements / DNA-templated DNA replication maintenance of fidelity / establishment of sister chromatid cohesion / maintenance of DNA repeat elements / DNA-templated DNA replication maintenance of fidelity /  gene conversion / Unwinding of DNA / replication fork arrest / Cul8-RING ubiquitin ligase complex / DNA replication initiation / meiotic chromosome segregation / epsilon DNA polymerase complex ... gene conversion / Unwinding of DNA / replication fork arrest / Cul8-RING ubiquitin ligase complex / DNA replication initiation / meiotic chromosome segregation / epsilon DNA polymerase complex ... establishment of sister chromatid cohesion / maintenance of DNA repeat elements / DNA-templated DNA replication maintenance of fidelity / establishment of sister chromatid cohesion / maintenance of DNA repeat elements / DNA-templated DNA replication maintenance of fidelity /  gene conversion / Unwinding of DNA / replication fork arrest / Cul8-RING ubiquitin ligase complex / DNA replication initiation / meiotic chromosome segregation / epsilon DNA polymerase complex / DNA strand elongation involved in mitotic DNA replication / MCM core complex / Assembly of the pre-replicative complex / Switching of origins to a post-replicative state / MCM complex binding / GINS complex / nuclear DNA replication / Termination of translesion DNA synthesis / mitotic DNA replication preinitiation complex assembly / premeiotic DNA replication / pre-replicative complex assembly involved in nuclear cell cycle DNA replication / gene conversion / Unwinding of DNA / replication fork arrest / Cul8-RING ubiquitin ligase complex / DNA replication initiation / meiotic chromosome segregation / epsilon DNA polymerase complex / DNA strand elongation involved in mitotic DNA replication / MCM core complex / Assembly of the pre-replicative complex / Switching of origins to a post-replicative state / MCM complex binding / GINS complex / nuclear DNA replication / Termination of translesion DNA synthesis / mitotic DNA replication preinitiation complex assembly / premeiotic DNA replication / pre-replicative complex assembly involved in nuclear cell cycle DNA replication /  SUMO binding / mitotic DNA replication / entrainment of circadian clock / Activation of the pre-replicative complex / CMG complex / establishment of mitotic sister chromatid cohesion / DNA replication checkpoint signaling / single-stranded DNA 3'-5' DNA exonuclease activity / nuclear pre-replicative complex / MCM complex / Activation of ATR in response to replication stress / DNA replication preinitiation complex / double-strand break repair via break-induced replication / replication fork protection complex / mitotic DNA replication initiation / mitotic DNA replication checkpoint signaling / single-stranded DNA helicase activity / nucleotide-excision repair, DNA gap filling / mitotic intra-S DNA damage checkpoint signaling / DNA replication proofreading / SUMO binding / mitotic DNA replication / entrainment of circadian clock / Activation of the pre-replicative complex / CMG complex / establishment of mitotic sister chromatid cohesion / DNA replication checkpoint signaling / single-stranded DNA 3'-5' DNA exonuclease activity / nuclear pre-replicative complex / MCM complex / Activation of ATR in response to replication stress / DNA replication preinitiation complex / double-strand break repair via break-induced replication / replication fork protection complex / mitotic DNA replication initiation / mitotic DNA replication checkpoint signaling / single-stranded DNA helicase activity / nucleotide-excision repair, DNA gap filling / mitotic intra-S DNA damage checkpoint signaling / DNA replication proofreading /  Hydrolases; Acting on ester bonds; Exodeoxyribonucleases producing 5'-phosphomonoesters / silent mating-type cassette heterochromatin formation / regulation of DNA-templated DNA replication initiation / mitotic sister chromatid cohesion / DNA strand elongation involved in DNA replication / nuclear chromosome / leading strand elongation / replication fork processing / DNA unwinding involved in DNA replication / nuclear replication fork / Hydrolases; Acting on ester bonds; Exodeoxyribonucleases producing 5'-phosphomonoesters / silent mating-type cassette heterochromatin formation / regulation of DNA-templated DNA replication initiation / mitotic sister chromatid cohesion / DNA strand elongation involved in DNA replication / nuclear chromosome / leading strand elongation / replication fork processing / DNA unwinding involved in DNA replication / nuclear replication fork /  DNA replication origin binding / Dual incision in TC-NER / subtelomeric heterochromatin formation / DNA replication initiation / error-prone translesion synthesis / heterochromatin formation / DNA replication origin binding / Dual incision in TC-NER / subtelomeric heterochromatin formation / DNA replication initiation / error-prone translesion synthesis / heterochromatin formation /  DNA helicase activity / DNA helicase activity /  base-excision repair, gap-filling / meiotic cell cycle / base-excision repair, gap-filling / meiotic cell cycle /  replication fork / replication fork /  helicase activity / helicase activity /  base-excision repair / DNA-templated DNA replication / double-strand break repair via nonhomologous end joining / double-strand break repair / base-excision repair / DNA-templated DNA replication / double-strand break repair via nonhomologous end joining / double-strand break repair /  nucleosome assembly / nucleosome assembly /  single-stranded DNA binding / mitotic cell cycle / 4 iron, 4 sulfur cluster binding / single-stranded DNA binding / mitotic cell cycle / 4 iron, 4 sulfur cluster binding /  double-stranded DNA binding / double-stranded DNA binding /  DNA helicase / DNA helicase /  DNA replication / DNA replication /  chromosome, telomeric region / chromosome, telomeric region /  DNA-directed DNA polymerase / DNA-directed DNA polymerase /  DNA-directed DNA polymerase activity / DNA-directed DNA polymerase activity /  cell cycle / cell cycle /  nucleotide binding / nucleotide binding /  DNA repair / DNA repair /  mRNA binding / DNA damage response / mRNA binding / DNA damage response /  chromatin binding / chromatin binding /  ATP hydrolysis activity / ATP hydrolysis activity /  DNA binding / zinc ion binding / DNA binding / zinc ion binding /  nucleoplasm / nucleoplasm /  ATP binding / identical protein binding / ATP binding / identical protein binding /  metal ion binding / metal ion binding /  nucleus / nucleus /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | |||||||||||||||||||||||||||
| Biological species |   Saccharomyces cerevisiae (brewer's yeast) / Saccharomyces cerevisiae (brewer's yeast) /   Saccharomyces cerevisiae S288C (yeast) Saccharomyces cerevisiae S288C (yeast) | |||||||||||||||||||||||||||
| Method |  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.07 Å cryo EM / Resolution: 3.07 Å | |||||||||||||||||||||||||||
 Authors Authors | Dang S / Zhai Y / Feng J / Yu D / Xu Z | |||||||||||||||||||||||||||
| Funding support |  Hong Kong, 8 items Hong Kong, 8 items
| |||||||||||||||||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2023 Journal: Nat Commun / Year: 2023Title: Synergism between CMG helicase and leading strand DNA polymerase at replication fork. Authors: Zhichun Xu / Jianrong Feng / Daqi Yu / Yunjing Huo / Xiaohui Ma / Wai Hei Lam / Zheng Liu / Xiang David Li / Toyotaka Ishibashi / Shangyu Dang / Yuanliang Zhai /  Abstract: The replisome that replicates the eukaryotic genome consists of at least three engines: the Cdc45-MCM-GINS (CMG) helicase that separates duplex DNA at the replication fork and two DNA polymerases, ...The replisome that replicates the eukaryotic genome consists of at least three engines: the Cdc45-MCM-GINS (CMG) helicase that separates duplex DNA at the replication fork and two DNA polymerases, one on each strand, that replicate the unwound DNA. Here, we determined a series of cryo-electron microscopy structures of a yeast replisome comprising CMG, leading-strand polymerase Polε and three accessory factors on a forked DNA. In these structures, Polε engages or disengages with the motor domains of the CMG by occupying two alternative positions, which closely correlate with the rotational movement of the single-stranded DNA around the MCM pore. During this process, the polymerase remains stably coupled to the helicase using Psf1 as a hinge. This synergism is modulated by a concerted rearrangement of ATPase sites to drive DNA translocation. The Polε-MCM coupling is not only required for CMG formation to initiate DNA replication but also facilitates the leading-strand DNA synthesis mediated by Polε. Our study elucidates a mechanism intrinsic to the replisome that coordinates the activities of CMG and Polε to negotiate any roadblocks, DNA damage, and epigenetic marks encountered during translocation along replication forks. | |||||||||||||||||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Supplemental images |
|---|
- Downloads & links
Downloads & links
-EMDB archive
| Map data |  emd_37211.map.gz emd_37211.map.gz | 567.3 MB |  EMDB map data format EMDB map data format | |
|---|---|---|---|---|
| Header (meta data) |  emd-37211-v30.xml emd-37211-v30.xml emd-37211.xml emd-37211.xml | 60.4 KB 60.4 KB | Display Display |  EMDB header EMDB header |
| Images |  emd_37211.png emd_37211.png | 118.6 KB | ||
| Masks |  emd_37211_msk_1.map emd_37211_msk_1.map | 600.7 MB |  Mask map Mask map | |
| Filedesc metadata |  emd-37211.cif.gz emd-37211.cif.gz | 16 KB | ||
| Others |  emd_37211_additional_1.map.gz emd_37211_additional_1.map.gz emd_37211_additional_2.map.gz emd_37211_additional_2.map.gz emd_37211_additional_3.map.gz emd_37211_additional_3.map.gz emd_37211_half_map_1.map.gz emd_37211_half_map_1.map.gz emd_37211_half_map_2.map.gz emd_37211_half_map_2.map.gz | 300.4 MB 300.6 MB 300.3 MB 556.9 MB 556.9 MB | ||
| Archive directory |  http://ftp.pdbj.org/pub/emdb/structures/EMD-37211 http://ftp.pdbj.org/pub/emdb/structures/EMD-37211 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37211 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-37211 | HTTPS FTP |
-Related structure data
| Related structure data |  8kg6MC  8kg8C  8kg9C  8w7mC  8w7sC C: citing same article ( M: atomic model generated by this map |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
| EMDB pages |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| Related items in Molecule of the Month |
- Map
Map
| File |  Download / File: emd_37211.map.gz / Format: CCP4 / Size: 600.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) Download / File: emd_37211.map.gz / Format: CCP4 / Size: 600.7 MB / Type: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Voxel size | X=Y=Z: 1.06 Å | ||||||||||||||||||||
| Density |
| ||||||||||||||||||||
| Symmetry | Space group: 1 | ||||||||||||||||||||
| Details | EMDB XML:
|
-Supplemental data
-Mask #1
| File |  emd_37211_msk_1.map emd_37211_msk_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Projections & Slices |
| ||||||||||||
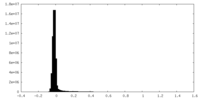
| Density Histograms |
-Additional map: focused refined map for DPB2/Pol2 modelling
| File | emd_37211_additional_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | focused refined map for DPB2/Pol2 modelling | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: A map for Csm3/Tof1 modelling
| File | emd_37211_additional_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | A map for Csm3/Tof1 modelling | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Additional map: A map for the MCM5 WH domain modelling
| File | emd_37211_additional_3.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | A map for the MCM5 WH domain modelling | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map1
| File | emd_37211_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map1 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
-Half map: Half map2
| File | emd_37211_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Annotation | Half map2 | ||||||||||||
| Projections & Slices |
| ||||||||||||
| Density Histograms |
- Sample components
Sample components
+Entire : replisome complex in state I
+Supramolecule #1: replisome complex in state I
+Macromolecule #1: DNA replication licensing factor MCM2
+Macromolecule #2: DNA replication licensing factor MCM3
+Macromolecule #3: DNA replication licensing factor MCM4
+Macromolecule #4: Minichromosome maintenance protein 5
+Macromolecule #5: DNA replication licensing factor MCM6
+Macromolecule #6: DNA replication licensing factor MCM7
+Macromolecule #7: DNA replication complex GINS protein PSF1
+Macromolecule #8: DNA replication complex GINS protein PSF2
+Macromolecule #9: DNA replication complex GINS protein PSF3
+Macromolecule #10: DNA replication complex GINS protein SLD5
+Macromolecule #11: Cell division control protein 45
+Macromolecule #12: DNA polymerase alpha-binding protein
+Macromolecule #15: Topoisomerase 1-associated factor 1
+Macromolecule #16: Chromosome segregation in meiosis protein 3
+Macromolecule #17: DNA polymerase epsilon catalytic subunit A
+Macromolecule #18: DNA polymerase epsilon subunit B
+Macromolecule #13: DNA (71-mer)
+Macromolecule #14: DNA (61-mer)
+Macromolecule #19: ZINC ION
+Macromolecule #20: ADENOSINE-5'-DIPHOSPHATE
+Macromolecule #21: MAGNESIUM ION
+Macromolecule #22: PHOSPHOTHIOPHOSPHORIC ACID-ADENYLATE ESTER
-Experimental details
-Structure determination
| Method |  cryo EM cryo EM |
|---|---|
 Processing Processing |  single particle reconstruction single particle reconstruction |
| Aggregation state | particle |
- Sample preparation
Sample preparation
| Concentration | 2 mg/mL | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Buffer | pH: 7.6 Component:
| |||||||||||||||
| Grid | Model: C-flat-1.2/1.3 / Material: GOLD / Mesh: 300 | |||||||||||||||
| Vitrification | Cryogen name: ETHANE / Chamber humidity: 100 % / Chamber temperature: 277.15 K / Instrument: FEI VITROBOT MARK IV Details: blot with filter paper for 3-4 seconds before plunging.. |
- Electron microscopy
Electron microscopy
| Microscope | FEI TITAN KRIOS |
|---|---|
| Electron beam | Acceleration voltage: 300 kV / Electron source:  FIELD EMISSION GUN FIELD EMISSION GUN |
| Electron optics | C2 aperture diameter: 100.0 µm / Illumination mode: FLOOD BEAM / Imaging mode: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 81000 Bright-field microscopy / Cs: 2.7 mm / Nominal defocus max: 2.5 µm / Nominal defocus min: 1.0 µm / Nominal magnification: 81000 |
| Specialist optics | Energy filter - Name: GIF Bioquantum / Energy filter - Slit width: 20 eV |
| Sample stage | Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER / Cooling holder cryogen: NITROGEN |
| Image recording | Film or detector model: GATAN K3 BIOQUANTUM (6k x 4k) / Digitization - Dimensions - Width: 5760 pixel / Digitization - Dimensions - Height: 4092 pixel / Number grids imaged: 4 / Number real images: 21776 / Average exposure time: 4.5 sec. / Average electron dose: 53.0 e/Å2 Details: Images were collected in movie-mode containing 40 frames. |
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
- Image processing
Image processing
| Startup model | Type of model: NONE |
|---|---|
| Initial angle assignment | Type: MAXIMUM LIKELIHOOD Software: (Name: cryoSPARC (ver. v3.0.1), cryoSPARC (ver. v2.15.0)) |
| Final angle assignment | Type: MAXIMUM LIKELIHOOD Software: (Name: cryoSPARC (ver. v3.0.1), cryoSPARC (ver. v2.15.0)) |
| Final reconstruction | Number classes used: 1 / Applied symmetry - Point group: C1 (asymmetric) / Algorithm: EXACT BACK PROJECTION / Resolution.type: BY AUTHOR / Resolution: 3.07 Å / Resolution method: FSC 0.143 CUT-OFF Software: (Name: cryoSPARC (ver. v3.0.1), cryoSPARC (ver. v2.15.0)) Number images used: 384519 |
 Movie
Movie Controller
Controller














 Z
Z Y
Y X
X