+Search query
-Structure paper
| Title | Cryo-EM reveals conformational flexibility in apo DNA polymerase ζ. |
|---|---|
| Journal, issue, pages | J Biol Chem, Vol. 297, Issue 2, Page 100912, Year 2021 |
| Publish date | Jun 24, 2021 |
 Authors Authors | Chloe Du Truong / Theodore A Craig / Gaofeng Cui / Maria Victoria Botuyan / Rachel A Serkasevich / Ka-Yi Chan / Georges Mer / Po-Lin Chiu / Rajiv Kumar /  |
| PubMed Abstract | The translesion synthesis (TLS) DNA polymerases Rev1 and Polζ function together in DNA lesion bypass during DNA replication, acting as nucleotide inserter and extender polymerases, respectively. ...The translesion synthesis (TLS) DNA polymerases Rev1 and Polζ function together in DNA lesion bypass during DNA replication, acting as nucleotide inserter and extender polymerases, respectively. While the structural characterization of the Saccharomyces cerevisiae Polζ in its DNA-bound state has illuminated how this enzyme synthesizes DNA, a mechanistic understanding of TLS also requires probing conformational changes associated with DNA- and Rev1 binding. Here, we used single-particle cryo-electron microscopy to determine the structure of the apo Polζ holoenzyme. We show that compared with its DNA-bound state, apo Polζ displays enhanced flexibility that correlates with concerted motions associated with expansion of the Polζ DNA-binding channel upon DNA binding. We also identified a lysine residue that obstructs the DNA-binding channel in apo Polζ, suggesting a gating mechanism. The Polζ subunit Rev7 is a hub protein that directly binds Rev1 and is a component of several other protein complexes such as the shieldin DNA double-strand break repair complex. We analyzed the molecular interactions of budding yeast Rev7 in the context of Polζ and those of human Rev7 in the context of shieldin using a crystal structure of Rev7 bound to a fragment of the shieldin-3 protein. Overall, our study provides new insights into Polζ mechanism of action and the manner in which Rev7 recognizes partner proteins. |
 External links External links |  J Biol Chem / J Biol Chem /  PubMed:34174285 / PubMed:34174285 /  PubMed Central PubMed Central |
| Methods | EM (single particle) / X-ray diffraction |
| Resolution | 2 - 4.11 Å |
| Structure data | EMDB-23570, PDB-7lxd:  PDB-6ve5: |
| Chemicals | 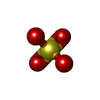 ChemComp-SO4:  ChemComp-HOH:  ChemComp-SF4: |
| Source |
|
 Keywords Keywords |  PROTEIN BINDING / DNA damage response / DNA double-strand break repair / DNA end resection / PROTEIN BINDING / DNA damage response / DNA double-strand break repair / DNA end resection /  homologous recombination / homologous recombination /  non-homologous end joining / Shieldin3 / SHLD3 / RINN1 / REV7 / non-homologous end joining / Shieldin3 / SHLD3 / RINN1 / REV7 /  53BP1 / RIF1 / 53BP1 / RIF1 /  DNA BINDING PROTEIN / DNA BINDING PROTEIN /  nucleic acid binding / nucleic acid binding /  DNA polymerase / DNA polymerase /  metal ion binding / metal ion binding /  catalytic activity catalytic activity |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers







