| 登録情報 | データベース: PDB / ID: 2pav
|
|---|
| タイトル | Ternary complex of Profilin-Actin with the Last Poly-Pro of Human VASP |
|---|
 要素 要素 | - Actin, alpha skeletal muscle
 - Profilin-1
 プロフィリン プロフィリン - Vasodilator-stimulated phosphoprotein
 血管拡張因子刺激リン酸化タンパク質 血管拡張因子刺激リン酸化タンパク質
|
|---|
 キーワード キーワード |  STRUCTURAL PROTEIN (タンパク質) / STRUCTURAL PROTEIN (タンパク質) /  Ternary complex / Ternary complex /  Profilin (プロフィリン) / Profilin (プロフィリン) /  Actin (アクチン) / Actin (アクチン) /  VASP (VASP航空) / Poly-Proline / Loading Poly-Pro Site VASP (VASP航空) / Poly-Proline / Loading Poly-Pro Site |
|---|
| 機能・相同性 |  機能・相同性情報 機能・相同性情報
synapse maturation / modification of postsynaptic actin cytoskeleton / negative regulation of actin filament bundle assembly / adenyl-nucleotide exchange factor activity /  profilin binding / positive regulation of actin filament bundle assembly / negative regulation of actin filament polymerization / Signaling by ROBO receptors / Cell-extracellular matrix interactions / regulation of actin filament polymerization ...synapse maturation / modification of postsynaptic actin cytoskeleton / negative regulation of actin filament bundle assembly / adenyl-nucleotide exchange factor activity / profilin binding / positive regulation of actin filament bundle assembly / negative regulation of actin filament polymerization / Signaling by ROBO receptors / Cell-extracellular matrix interactions / regulation of actin filament polymerization ...synapse maturation / modification of postsynaptic actin cytoskeleton / negative regulation of actin filament bundle assembly / adenyl-nucleotide exchange factor activity /  profilin binding / positive regulation of actin filament bundle assembly / negative regulation of actin filament polymerization / Signaling by ROBO receptors / Cell-extracellular matrix interactions / regulation of actin filament polymerization / actin polymerization or depolymerization / positive regulation of ATP-dependent activity / PCP/CE pathway / filopodium membrane / proline-rich region binding / positive regulation of ruffle assembly / negative regulation of stress fiber assembly / cytoskeletal motor activator activity / positive regulation of actin filament polymerization / positive regulation of epithelial cell migration / profilin binding / positive regulation of actin filament bundle assembly / negative regulation of actin filament polymerization / Signaling by ROBO receptors / Cell-extracellular matrix interactions / regulation of actin filament polymerization / actin polymerization or depolymerization / positive regulation of ATP-dependent activity / PCP/CE pathway / filopodium membrane / proline-rich region binding / positive regulation of ruffle assembly / negative regulation of stress fiber assembly / cytoskeletal motor activator activity / positive regulation of actin filament polymerization / positive regulation of epithelial cell migration /  tropomyosin binding / tropomyosin binding /  myosin heavy chain binding / mesenchyme migration / myosin heavy chain binding / mesenchyme migration /  troponin I binding / actin filament bundle / filamentous actin / lamellipodium membrane / skeletal muscle thin filament assembly / actin filament bundle assembly / striated muscle thin filament / Generation of second messenger molecules / skeletal muscle myofibril / actin monomer binding / bicellular tight junction / skeletal muscle fiber development / troponin I binding / actin filament bundle / filamentous actin / lamellipodium membrane / skeletal muscle thin filament assembly / actin filament bundle assembly / striated muscle thin filament / Generation of second messenger molecules / skeletal muscle myofibril / actin monomer binding / bicellular tight junction / skeletal muscle fiber development /  stress fiber / stress fiber /  titin binding / titin binding /  phosphatidylinositol-4,5-bisphosphate binding / actin filament polymerization / phosphotyrosine residue binding / phosphatidylinositol-4,5-bisphosphate binding / actin filament polymerization / phosphotyrosine residue binding /  filopodium / neural tube closure / RHO GTPases Activate Formins / filopodium / neural tube closure / RHO GTPases Activate Formins /  マイクロフィラメント / マイクロフィラメント /  軸索誘導 / 軸索誘導 /  加水分解酵素; 酸無水物に作用; 酸無水物に作用・細胞または細胞小器官の運動に関与 / modulation of chemical synaptic transmission / 加水分解酵素; 酸無水物に作用; 酸無水物に作用・細胞または細胞小器官の運動に関与 / modulation of chemical synaptic transmission /  small GTPase binding / small GTPase binding /  SH3 domain binding / calcium-dependent protein binding / SH3 domain binding / calcium-dependent protein binding /  マイクロフィラメント / Platelet degranulation / マイクロフィラメント / Platelet degranulation /  lamellipodium / lamellipodium /  cell body / cell body /  actin binding / actin binding /  細胞皮質 / actin cytoskeleton organization / protein homotetramerization / blood microparticle / protein stabilization / 細胞皮質 / actin cytoskeleton organization / protein homotetramerization / blood microparticle / protein stabilization /  細胞骨格 / 細胞骨格 /  hydrolase activity / hydrolase activity /  cadherin binding / protein domain specific binding / cadherin binding / protein domain specific binding /  focal adhesion / glutamatergic synapse / focal adhesion / glutamatergic synapse /  calcium ion binding / positive regulation of gene expression / regulation of transcription by RNA polymerase II / magnesium ion binding / calcium ion binding / positive regulation of gene expression / regulation of transcription by RNA polymerase II / magnesium ion binding /  RNA binding / extracellular exosome / RNA binding / extracellular exosome /  ATP binding / ATP binding /  生体膜 / identical protein binding / 生体膜 / identical protein binding /  細胞核 / 細胞核 /  細胞膜 / 細胞膜 /  細胞質基質 / 細胞質基質 /  細胞質類似検索 - 分子機能 細胞質類似検索 - 分子機能 Vasodilator-stimulated phosphoprotein/ENA/VASP-like / Profilin1/2/3, vertebrate / VASP tetramerisation / VASP tetramerisation domain superfamily / VASP tetramerisation domain / : / Profilin conserved site / Profilin signature. / WH1/EVH1 domain /  WH1 domain ...Vasodilator-stimulated phosphoprotein/ENA/VASP-like / Profilin1/2/3, vertebrate / VASP tetramerisation / VASP tetramerisation domain superfamily / VASP tetramerisation domain / : / Profilin conserved site / Profilin signature. / WH1/EVH1 domain / WH1 domain ...Vasodilator-stimulated phosphoprotein/ENA/VASP-like / Profilin1/2/3, vertebrate / VASP tetramerisation / VASP tetramerisation domain superfamily / VASP tetramerisation domain / : / Profilin conserved site / Profilin signature. / WH1/EVH1 domain /  WH1 domain / WH1 domain profile. / WASP homology region 1 / WH1 domain / WH1 domain profile. / WASP homology region 1 /  プロフィリン / プロフィリン /  プロフィリン / プロフィリン /  プロフィリン / Profilin superfamily / Dynein light chain 2a, cytoplasmic / プロフィリン / Profilin superfamily / Dynein light chain 2a, cytoplasmic /  ATPase, substrate binding domain, subdomain 4 / ATPase, substrate binding domain, subdomain 4 /  Actin; Chain A, domain 4 / Actin; Chain A, domain 4 /  ATPase, nucleotide binding domain / Actins signature 1. / ATPase, nucleotide binding domain / Actins signature 1. /  Actin, conserved site / Actins signature 2. / Actin/actin-like conserved site / Actins and actin-related proteins signature. / Actin, conserved site / Actins signature 2. / Actin/actin-like conserved site / Actins and actin-related proteins signature. /  Β-ラクタマーゼ / Β-ラクタマーゼ /  アクチン / アクチン /  Actin family / Actin family /  アクチン / アクチン /  ATPase, nucleotide binding domain / ATPase, nucleotide binding domain /  Nucleotidyltransferase; domain 5 / PH-like domain superfamily / Nucleotidyltransferase; domain 5 / PH-like domain superfamily /  Alpha-Beta Complex / 2-Layer Sandwich / Alpha Beta類似検索 - ドメイン・相同性 Alpha-Beta Complex / 2-Layer Sandwich / Alpha Beta類似検索 - ドメイン・相同性 ADENOSINE-5'-TRIPHOSPHATE /  Profilin-1 / Profilin-1 /  血管拡張因子刺激リン酸化タンパク質 / 血管拡張因子刺激リン酸化タンパク質 /  Actin, alpha skeletal muscle類似検索 - 構成要素 Actin, alpha skeletal muscle類似検索 - 構成要素 |
|---|
| 生物種 |   Homo sapiens (ヒト) Homo sapiens (ヒト)
   Oryctolagus cuniculus (ウサギ) Oryctolagus cuniculus (ウサギ) |
|---|
| 手法 |  X線回折 / X線回折 /  シンクロトロン / シンクロトロン /  分子置換 / 解像度: 1.8 Å 分子置換 / 解像度: 1.8 Å |
|---|
 データ登録者 データ登録者 | Ferron, F. / Rebowski, G. / Dominguez, R. |
|---|
 引用 引用 |  ジャーナル: Embo J. / 年: 2007 ジャーナル: Embo J. / 年: 2007
タイトル: Structural basis for the recruitment of profilin-actin complexes during filament elongation by Ena/VASP.
著者: Ferron, F. / Rebowski, G. / Lee, S.H. / Dominguez, R. |
|---|
| 履歴 | | 登録 | 2007年3月27日 | 登録サイト: RCSB / 処理サイト: RCSB |
|---|
| 改定 1.0 | 2007年10月23日 | Provider: repository / タイプ: Initial release |
|---|
| 改定 1.1 | 2011年7月13日 | Group: Version format compliance |
|---|
| 改定 1.2 | 2013年3月6日 | Group: Advisory / Other |
|---|
| 改定 1.3 | 2023年8月30日 | Group: Data collection / Database references ...Data collection / Database references / Derived calculations / Refinement description
カテゴリ: chem_comp_atom / chem_comp_bond ...chem_comp_atom / chem_comp_bond / database_2 / pdbx_initial_refinement_model / pdbx_struct_conn_angle / struct_conn / struct_conn_type / struct_ref_seq_dif / struct_sheet / struct_site
Item: _database_2.pdbx_DOI / _database_2.pdbx_database_accession ..._database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.value / _struct_conn.conn_type_id / _struct_conn.id / _struct_conn.pdbx_dist_value / _struct_conn.pdbx_leaving_atom_flag / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id / _struct_conn_type.id / _struct_ref_seq_dif.details / _struct_sheet.number_strands / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id |
|---|
|
|---|
 データを開く
データを開く 基本情報
基本情報 要素
要素 キーワード
キーワード STRUCTURAL PROTEIN (タンパク質) /
STRUCTURAL PROTEIN (タンパク質) /  Ternary complex /
Ternary complex /  Profilin (プロフィリン) /
Profilin (プロフィリン) /  Actin (アクチン) /
Actin (アクチン) /  VASP (VASP航空) / Poly-Proline / Loading Poly-Pro Site
VASP (VASP航空) / Poly-Proline / Loading Poly-Pro Site 機能・相同性情報
機能・相同性情報 profilin binding / positive regulation of actin filament bundle assembly / negative regulation of actin filament polymerization / Signaling by ROBO receptors / Cell-extracellular matrix interactions / regulation of actin filament polymerization ...synapse maturation / modification of postsynaptic actin cytoskeleton / negative regulation of actin filament bundle assembly / adenyl-nucleotide exchange factor activity /
profilin binding / positive regulation of actin filament bundle assembly / negative regulation of actin filament polymerization / Signaling by ROBO receptors / Cell-extracellular matrix interactions / regulation of actin filament polymerization ...synapse maturation / modification of postsynaptic actin cytoskeleton / negative regulation of actin filament bundle assembly / adenyl-nucleotide exchange factor activity /  profilin binding / positive regulation of actin filament bundle assembly / negative regulation of actin filament polymerization / Signaling by ROBO receptors / Cell-extracellular matrix interactions / regulation of actin filament polymerization / actin polymerization or depolymerization / positive regulation of ATP-dependent activity / PCP/CE pathway / filopodium membrane / proline-rich region binding / positive regulation of ruffle assembly / negative regulation of stress fiber assembly / cytoskeletal motor activator activity / positive regulation of actin filament polymerization / positive regulation of epithelial cell migration /
profilin binding / positive regulation of actin filament bundle assembly / negative regulation of actin filament polymerization / Signaling by ROBO receptors / Cell-extracellular matrix interactions / regulation of actin filament polymerization / actin polymerization or depolymerization / positive regulation of ATP-dependent activity / PCP/CE pathway / filopodium membrane / proline-rich region binding / positive regulation of ruffle assembly / negative regulation of stress fiber assembly / cytoskeletal motor activator activity / positive regulation of actin filament polymerization / positive regulation of epithelial cell migration /  tropomyosin binding /
tropomyosin binding /  myosin heavy chain binding / mesenchyme migration /
myosin heavy chain binding / mesenchyme migration /  troponin I binding / actin filament bundle / filamentous actin / lamellipodium membrane / skeletal muscle thin filament assembly / actin filament bundle assembly / striated muscle thin filament / Generation of second messenger molecules / skeletal muscle myofibril / actin monomer binding / bicellular tight junction / skeletal muscle fiber development /
troponin I binding / actin filament bundle / filamentous actin / lamellipodium membrane / skeletal muscle thin filament assembly / actin filament bundle assembly / striated muscle thin filament / Generation of second messenger molecules / skeletal muscle myofibril / actin monomer binding / bicellular tight junction / skeletal muscle fiber development /  stress fiber /
stress fiber /  titin binding /
titin binding /  phosphatidylinositol-4,5-bisphosphate binding / actin filament polymerization / phosphotyrosine residue binding /
phosphatidylinositol-4,5-bisphosphate binding / actin filament polymerization / phosphotyrosine residue binding /  filopodium / neural tube closure / RHO GTPases Activate Formins /
filopodium / neural tube closure / RHO GTPases Activate Formins /  マイクロフィラメント /
マイクロフィラメント /  軸索誘導 /
軸索誘導 /  加水分解酵素; 酸無水物に作用; 酸無水物に作用・細胞または細胞小器官の運動に関与 / modulation of chemical synaptic transmission /
加水分解酵素; 酸無水物に作用; 酸無水物に作用・細胞または細胞小器官の運動に関与 / modulation of chemical synaptic transmission /  small GTPase binding /
small GTPase binding /  SH3 domain binding / calcium-dependent protein binding /
SH3 domain binding / calcium-dependent protein binding /  マイクロフィラメント / Platelet degranulation /
マイクロフィラメント / Platelet degranulation /  lamellipodium /
lamellipodium /  cell body /
cell body /  actin binding /
actin binding /  細胞皮質 / actin cytoskeleton organization / protein homotetramerization / blood microparticle / protein stabilization /
細胞皮質 / actin cytoskeleton organization / protein homotetramerization / blood microparticle / protein stabilization /  細胞骨格 /
細胞骨格 /  hydrolase activity /
hydrolase activity /  cadherin binding / protein domain specific binding /
cadherin binding / protein domain specific binding /  focal adhesion / glutamatergic synapse /
focal adhesion / glutamatergic synapse /  calcium ion binding / positive regulation of gene expression / regulation of transcription by RNA polymerase II / magnesium ion binding /
calcium ion binding / positive regulation of gene expression / regulation of transcription by RNA polymerase II / magnesium ion binding /  RNA binding / extracellular exosome /
RNA binding / extracellular exosome /  ATP binding /
ATP binding /  生体膜 / identical protein binding /
生体膜 / identical protein binding /  細胞核 /
細胞核 /  細胞膜 /
細胞膜 /  細胞質基質 /
細胞質基質 /  細胞質
細胞質
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Oryctolagus cuniculus (ウサギ)
Oryctolagus cuniculus (ウサギ) X線回折 /
X線回折 /  シンクロトロン /
シンクロトロン /  分子置換 / 解像度: 1.8 Å
分子置換 / 解像度: 1.8 Å  データ登録者
データ登録者 引用
引用 ジャーナル: Embo J. / 年: 2007
ジャーナル: Embo J. / 年: 2007 構造の表示
構造の表示 Molmil
Molmil Jmol/JSmol
Jmol/JSmol ダウンロードとリンク
ダウンロードとリンク ダウンロード
ダウンロード 2pav.cif.gz
2pav.cif.gz PDBx/mmCIF形式
PDBx/mmCIF形式 pdb2pav.ent.gz
pdb2pav.ent.gz PDB形式
PDB形式 2pav.json.gz
2pav.json.gz PDBx/mmJSON形式
PDBx/mmJSON形式 その他のダウンロード
その他のダウンロード https://data.pdbj.org/pub/pdb/validation_reports/pa/2pav
https://data.pdbj.org/pub/pdb/validation_reports/pa/2pav ftp://data.pdbj.org/pub/pdb/validation_reports/pa/2pav
ftp://data.pdbj.org/pub/pdb/validation_reports/pa/2pav リンク
リンク 集合体
集合体
 要素
要素 / Alpha-actin-1
/ Alpha-actin-1
 Oryctolagus cuniculus (ウサギ) / 参照: UniProt: P68135
Oryctolagus cuniculus (ウサギ) / 参照: UniProt: P68135 プロフィリン / Profilin I
プロフィリン / Profilin I
 Homo sapiens (ヒト) / 遺伝子: PFN1 / プラスミド: pET29 / 生物種 (発現宿主): Escherichia coli / 発現宿主:
Homo sapiens (ヒト) / 遺伝子: PFN1 / プラスミド: pET29 / 生物種 (発現宿主): Escherichia coli / 発現宿主: 
 Escherichia coli BL21(DE3) (大腸菌) / 株 (発現宿主): BL21(DE3) / 参照: UniProt: P07737
Escherichia coli BL21(DE3) (大腸菌) / 株 (発現宿主): BL21(DE3) / 参照: UniProt: P07737 血管拡張因子刺激リン酸化タンパク質 / VASP
血管拡張因子刺激リン酸化タンパク質 / VASP
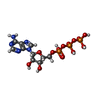



 アデノシン三リン酸
アデノシン三リン酸 水
水 X線回折 / 使用した結晶の数: 1
X線回折 / 使用した結晶の数: 1  試料調製
試料調製
 シンクロトロン / サイト:
シンクロトロン / サイト:  APS
APS  / ビームライン: 14-BM-C / 波長: 0.9002 Å
/ ビームライン: 14-BM-C / 波長: 0.9002 Å : 0.9002 Å / 相対比: 1
: 0.9002 Å / 相対比: 1  解析
解析 :
:  分子置換
分子置換 ムービー
ムービー コントローラー
コントローラー















 PDBj
PDBj














