+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1pa2 | ||||||
|---|---|---|---|---|---|---|---|
| Title | ARABIDOPSIS THALIANA PEROXIDASE A2 | ||||||
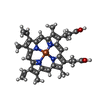
 Components Components | PEROXIDASE | ||||||
 Keywords Keywords |  OXIDOREDUCTASE / OXIDOREDUCTASE /  PEROXIDASE PEROXIDASE | ||||||
| Function / homology |  Function and homology information Function and homology informationdefense response to nematode /  flower development / flower development /  lactoperoxidase activity / lactoperoxidase activity /  peroxidase / hydrogen peroxide catabolic process / response to oxidative stress / peroxidase / hydrogen peroxide catabolic process / response to oxidative stress /  heme binding / heme binding /  Golgi apparatus / extracellular region / Golgi apparatus / extracellular region /  metal ion binding metal ion bindingSimilarity search - Function | ||||||
| Biological species |   Arabidopsis thaliana (thale cress) Arabidopsis thaliana (thale cress) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.45 Å MOLECULAR REPLACEMENT / Resolution: 1.45 Å | ||||||
 Authors Authors | Henriksen, A. | ||||||
 Citation Citation |  Journal: Plant Mol.Biol. / Year: 2000 Journal: Plant Mol.Biol. / Year: 2000Title: Arabidopsis ATP A2 peroxidase. Expression and high-resolution structure of a plant peroxidase with implications for lignification. Authors: Ostergaard, L. / Teilum, K. / Mirza, O. / Mattsson, O. / Petersen, M. / Welinder, K.G. / Mundy, J. / Gajhede, M. / Henriksen, A. #1:  Journal: FEBS Lett. / Year: 1996 Journal: FEBS Lett. / Year: 1996Title: Structure and Organ Specificity of an Anionic Peroxidase from Arabidopsis Thaliana Cell Suspension Culture Authors: Ostergaard, L. / Abelskov, A.K. / Mattsson, O. / Welinder, K.G. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1pa2.cif.gz 1pa2.cif.gz | 122.1 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1pa2.ent.gz pdb1pa2.ent.gz | 87.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1pa2.json.gz 1pa2.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/pa/1pa2 https://data.pdbj.org/pub/pdb/validation_reports/pa/1pa2 ftp://data.pdbj.org/pub/pdb/validation_reports/pa/1pa2 ftp://data.pdbj.org/pub/pdb/validation_reports/pa/1pa2 | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  1atjS S: Starting model for refinement |
|---|---|
| Similar structure data |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein |  / ATP A2 / ATP A2Mass: 32085.516 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Arabidopsis thaliana (thale cress) / Production host: Arabidopsis thaliana (thale cress) / Production host:   Escherichia coli (E. coli) / References: UniProt: Q42578, Escherichia coli (E. coli) / References: UniProt: Q42578,  peroxidase peroxidase | ||||||
|---|---|---|---|---|---|---|---|
| #2: Chemical | | #3: Chemical | ChemComp-MG / | #4: Chemical | ChemComp-HEM / |  Heme B Heme B#5: Water | ChemComp-HOH / |  Water Water |
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.16 Å3/Da / Density % sol: 42.99 % | ||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Crystal grow | pH: 6.5 / Details: pH 6.5 | ||||||||||||||||||||||||||||||||||||||||
| Crystal grow | *PLUS Method: vapor diffusion, hanging drop | ||||||||||||||||||||||||||||||||||||||||
| Components of the solutions | *PLUS
|
-Data collection
| Diffraction | Mean temperature: 100 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  MAX II MAX II  / Beamline: I711 / Wavelength: 0.97 / Beamline: I711 / Wavelength: 0.97 |
| Detector | Type: MARRESEARCH / Detector: IMAGE PLATE / Date: May 1, 1998 / Details: VERTICALLY FOCUSING CYLINDRICAL MIRROR |
| Radiation | Monochromator: SI(111) / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.97 Å / Relative weight: 1 : 0.97 Å / Relative weight: 1 |
| Reflection | Resolution: 1.45→99 Å / Num. obs: 49745 / % possible obs: 99 % / Observed criterion σ(I): 0 / Redundancy: 5.6 % / Rsym value: 0.058 / Net I/σ(I): 19.1 |
| Reflection shell | Resolution: 1.45→1.51 Å / Redundancy: 4 % / Mean I/σ(I) obs: 8.4 / Rsym value: 0.202 / % possible all: 99 |
| Reflection | *PLUS Lowest resolution: 99 Å / % possible obs: 99 % / Num. measured all: 280003 / Rmerge(I) obs: 0.058 |
| Reflection shell | *PLUS % possible obs: 99 % / Num. unique obs: 5403 / Rmerge(I) obs: 0.243 / Mean I/σ(I) obs: 6.4 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 1ATJ Resolution: 1.45→99 Å / Num. parameters: 11562 / Num. restraintsaints: 9824 / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: ENGH AND HUBER
| |||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: MOEWS & KRETSINGER, J.MOL.BIOL.91(1973)201-2 | |||||||||||||||||||||||||||||||||
| Refine analyze | Num. disordered residues: 29 / Occupancy sum hydrogen: 2187 / Occupancy sum non hydrogen: 2775 | |||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.45→99 Å
| |||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||
| Software | *PLUS Name: SHELXL-96 / Classification: refinement | |||||||||||||||||||||||||||||||||
| Refinement | *PLUS Lowest resolution: 99 Å / σ(F): 4 / % reflection Rfree: 5 % / Rfactor all : 0.1477 / Rfactor obs: 0.1398 : 0.1477 / Rfactor obs: 0.1398 | |||||||||||||||||||||||||||||||||
| Solvent computation | *PLUS | |||||||||||||||||||||||||||||||||
| Displacement parameters | *PLUS |
 Movie
Movie Controller
Controller













 PDBj
PDBj