+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Proteasome-ZFAND5 Complex Z+E state | |||||||||
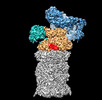
 マップデータ マップデータ | ZFAND5-proteasome complex Z E state | |||||||||
 試料 試料 |
| |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報Impaired BRCA2 translocation to the nucleus / Impaired BRCA2 binding to SEM1 (DSS1) / modulation by host of viral transcription /  加水分解酵素; プロテアーゼ; ペプチド結合加水分解酵素; オメガペプチターゼ / proteasome accessory complex / 加水分解酵素; プロテアーゼ; ペプチド結合加水分解酵素; オメガペプチターゼ / proteasome accessory complex /  減数分裂 / purine ribonucleoside triphosphate binding / positive regulation of proteasomal protein catabolic process / metal-dependent deubiquitinase activity / proteasome regulatory particle ...Impaired BRCA2 translocation to the nucleus / Impaired BRCA2 binding to SEM1 (DSS1) / modulation by host of viral transcription / 減数分裂 / purine ribonucleoside triphosphate binding / positive regulation of proteasomal protein catabolic process / metal-dependent deubiquitinase activity / proteasome regulatory particle ...Impaired BRCA2 translocation to the nucleus / Impaired BRCA2 binding to SEM1 (DSS1) / modulation by host of viral transcription /  加水分解酵素; プロテアーゼ; ペプチド結合加水分解酵素; オメガペプチターゼ / proteasome accessory complex / 加水分解酵素; プロテアーゼ; ペプチド結合加水分解酵素; オメガペプチターゼ / proteasome accessory complex /  減数分裂 / purine ribonucleoside triphosphate binding / positive regulation of proteasomal protein catabolic process / metal-dependent deubiquitinase activity / proteasome regulatory particle / cytosolic proteasome complex / proteasome regulatory particle, lid subcomplex / proteasome-activating activity / proteasome regulatory particle, base subcomplex / protein K63-linked deubiquitination / 減数分裂 / purine ribonucleoside triphosphate binding / positive regulation of proteasomal protein catabolic process / metal-dependent deubiquitinase activity / proteasome regulatory particle / cytosolic proteasome complex / proteasome regulatory particle, lid subcomplex / proteasome-activating activity / proteasome regulatory particle, base subcomplex / protein K63-linked deubiquitination /  regulation of endopeptidase activity / Defective homologous recombination repair (HRR) due to BRCA1 loss of function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA1 binding function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA2/RAD51/RAD51C binding function / Regulation of ornithine decarboxylase (ODC) / proteasome core complex / Homologous DNA Pairing and Strand Exchange / Resolution of D-loop Structures through Synthesis-Dependent Strand Annealing (SDSA) / Resolution of D-loop Structures through Holliday Junction Intermediates / Cross-presentation of soluble exogenous antigens (endosomes) / regulation of endopeptidase activity / Defective homologous recombination repair (HRR) due to BRCA1 loss of function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA1 binding function / Defective HDR through Homologous Recombination Repair (HRR) due to PALB2 loss of BRCA2/RAD51/RAD51C binding function / Regulation of ornithine decarboxylase (ODC) / proteasome core complex / Homologous DNA Pairing and Strand Exchange / Resolution of D-loop Structures through Synthesis-Dependent Strand Annealing (SDSA) / Resolution of D-loop Structures through Holliday Junction Intermediates / Cross-presentation of soluble exogenous antigens (endosomes) /  Somitogenesis / K63-linked deubiquitinase activity / Impaired BRCA2 binding to RAD51 / Somitogenesis / K63-linked deubiquitinase activity / Impaired BRCA2 binding to RAD51 /  immune system process / immune system process /  myofibril / myofibril /  proteasome binding / regulation of protein catabolic process / proteasome storage granule / Presynaptic phase of homologous DNA pairing and strand exchange / blastocyst development / polyubiquitin modification-dependent protein binding / endopeptidase activator activity / proteasome binding / regulation of protein catabolic process / proteasome storage granule / Presynaptic phase of homologous DNA pairing and strand exchange / blastocyst development / polyubiquitin modification-dependent protein binding / endopeptidase activator activity /  NF-kappaB binding / NF-kappaB binding /  proteasome assembly / proteasome assembly /  proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / proteasome core complex, alpha-subunit complex / threonine-type endopeptidase activity / enzyme regulator activity / mRNA export from nucleus / regulation of proteasomal protein catabolic process / negative regulation of inflammatory response to antigenic stimulus / response to organonitrogen compound / proteasome endopeptidase complex / proteasome core complex, beta-subunit complex / proteasome core complex, alpha-subunit complex / threonine-type endopeptidase activity / enzyme regulator activity / mRNA export from nucleus / regulation of proteasomal protein catabolic process / negative regulation of inflammatory response to antigenic stimulus / response to organonitrogen compound /  proteasome complex / proteolysis involved in protein catabolic process / proteasome complex / proteolysis involved in protein catabolic process /  sarcomere / Regulation of activated PAK-2p34 by proteasome mediated degradation / ciliary basal body / N-glycan trimming in the ER and Calnexin/Calreticulin cycle / Autodegradation of Cdh1 by Cdh1:APC/C / APC/C:Cdc20 mediated degradation of Securin / Asymmetric localization of PCP proteins / SCF-beta-TrCP mediated degradation of Emi1 / NIK-->noncanonical NF-kB signaling / Ubiquitin-dependent degradation of Cyclin D / AUF1 (hnRNP D0) binds and destabilizes mRNA / TNFR2 non-canonical NF-kB pathway / Assembly of the pre-replicative complex / Vpu mediated degradation of CD4 / proteasomal protein catabolic process / Degradation of DVL / sarcomere / Regulation of activated PAK-2p34 by proteasome mediated degradation / ciliary basal body / N-glycan trimming in the ER and Calnexin/Calreticulin cycle / Autodegradation of Cdh1 by Cdh1:APC/C / APC/C:Cdc20 mediated degradation of Securin / Asymmetric localization of PCP proteins / SCF-beta-TrCP mediated degradation of Emi1 / NIK-->noncanonical NF-kB signaling / Ubiquitin-dependent degradation of Cyclin D / AUF1 (hnRNP D0) binds and destabilizes mRNA / TNFR2 non-canonical NF-kB pathway / Assembly of the pre-replicative complex / Vpu mediated degradation of CD4 / proteasomal protein catabolic process / Degradation of DVL /  P-body / P-body /  細胞分化 / Ubiquitin Mediated Degradation of Phosphorylated Cdc25A / Dectin-1 mediated noncanonical NF-kB signaling / Hh mutants are degraded by ERAD / Cdc20:Phospho-APC/C mediated degradation of Cyclin A / Degradation of AXIN / Defective CFTR causes cystic fibrosis / Degradation of GLI1 by the proteasome / 細胞分化 / Ubiquitin Mediated Degradation of Phosphorylated Cdc25A / Dectin-1 mediated noncanonical NF-kB signaling / Hh mutants are degraded by ERAD / Cdc20:Phospho-APC/C mediated degradation of Cyclin A / Degradation of AXIN / Defective CFTR causes cystic fibrosis / Degradation of GLI1 by the proteasome /  lipopolysaccharide binding / Hedgehog ligand biogenesis / Activation of NF-kappaB in B cells / Negative regulation of NOTCH4 signaling / GSK3B and BTRC:CUL1-mediated-degradation of NFE2L2 / G2/M Checkpoints / double-strand break repair via homologous recombination / Vif-mediated degradation of APOBEC3G / Autodegradation of the E3 ubiquitin ligase COP1 / Hedgehog 'on' state / Regulation of RUNX3 expression and activity / Degradation of GLI2 by the proteasome / GLI3 is processed to GLI3R by the proteasome / MAPK6/MAPK4 signaling / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / response to virus / APC/C:Cdh1 mediated degradation of Cdc20 and other APC/C:Cdh1 targeted proteins in late mitosis/early G1 / ABC-family proteins mediated transport / Degradation of beta-catenin by the destruction complex / Oxygen-dependent proline hydroxylation of Hypoxia-inducible Factor Alpha / HDR through Homologous Recombination (HRR) / cytoplasmic ribonucleoprotein granule / response to organic cyclic compound / Metalloprotease DUBs / CDK-mediated phosphorylation and removal of Cdc6 lipopolysaccharide binding / Hedgehog ligand biogenesis / Activation of NF-kappaB in B cells / Negative regulation of NOTCH4 signaling / GSK3B and BTRC:CUL1-mediated-degradation of NFE2L2 / G2/M Checkpoints / double-strand break repair via homologous recombination / Vif-mediated degradation of APOBEC3G / Autodegradation of the E3 ubiquitin ligase COP1 / Hedgehog 'on' state / Regulation of RUNX3 expression and activity / Degradation of GLI2 by the proteasome / GLI3 is processed to GLI3R by the proteasome / MAPK6/MAPK4 signaling / FBXL7 down-regulates AURKA during mitotic entry and in early mitosis / response to virus / APC/C:Cdh1 mediated degradation of Cdc20 and other APC/C:Cdh1 targeted proteins in late mitosis/early G1 / ABC-family proteins mediated transport / Degradation of beta-catenin by the destruction complex / Oxygen-dependent proline hydroxylation of Hypoxia-inducible Factor Alpha / HDR through Homologous Recombination (HRR) / cytoplasmic ribonucleoprotein granule / response to organic cyclic compound / Metalloprotease DUBs / CDK-mediated phosphorylation and removal of Cdc6類似検索 - 分子機能 | |||||||||
| 生物種 |   Homo sapiens (ヒト) / Homo sapiens (ヒト) /   human (ヒト) human (ヒト) | |||||||||
| 手法 |  単粒子再構成法 / 単粒子再構成法 /  クライオ電子顕微鏡法 / 解像度: 4.4 Å クライオ電子顕微鏡法 / 解像度: 4.4 Å | |||||||||
 データ登録者 データ登録者 | Zhu Y / Lu Y | |||||||||
| 資金援助 | 1件
| |||||||||
 引用 引用 |  ジャーナル: To Be Published ジャーナル: To Be Publishedタイトル: Mechanism of 26S proteasome activation by the 19S-interacting protein ZFAND5 著者: Zhu Y / Lu Y | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_14205.map.gz emd_14205.map.gz | 114.5 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-14205-v30.xml emd-14205-v30.xml emd-14205.xml emd-14205.xml | 43.1 KB 43.1 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_14205.png emd_14205.png | 116.1 KB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-14205 http://ftp.pdbj.org/pub/emdb/structures/EMD-14205 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14205 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-14205 | HTTPS FTP |
-関連構造データ
| 関連構造データ |  7qxxMC  7qxnC  7qxpC  7qxuC  7qxwC  7qy7C  7qyaC  7qybC M: このマップから作成された原子モデル C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_14205.map.gz / 形式: CCP4 / 大きさ: 125 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_14205.map.gz / 形式: CCP4 / 大きさ: 125 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | ZFAND5-proteasome complex Z E state | ||||||||||||||||||||||||||||||||||||
| 投影像・断面図 | 画像のコントロール
画像は Spider により作成 | ||||||||||||||||||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 1.37 Å | ||||||||||||||||||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
- 試料の構成要素
試料の構成要素
+全体 : human proteasome zfand5 complex Z+B state
+超分子 #1: human proteasome zfand5 complex Z+B state
+分子 #1: 26S proteasome non-ATPase regulatory subunit 1
+分子 #2: 26S proteasome non-ATPase regulatory subunit 3
+分子 #3: 26S proteasome non-ATPase regulatory subunit 12
+分子 #4: 26S proteasome non-ATPase regulatory subunit 11
+分子 #5: 26S proteasome non-ATPase regulatory subunit 6
+分子 #6: 26S proteasome non-ATPase regulatory subunit 7
+分子 #7: 26S proteasome non-ATPase regulatory subunit 13
+分子 #8: 26S proteasome non-ATPase regulatory subunit 4
+分子 #9: 26S proteasome non-ATPase regulatory subunit 14
+分子 #10: 26S proteasome non-ATPase regulatory subunit 8
+分子 #11: 26S proteasome complex subunit SEM1
+分子 #12: 26S proteasome non-ATPase regulatory subunit 2
+分子 #13: 26S protease regulatory subunit 7
+分子 #14: 26S proteasome regulatory subunit 4
+分子 #15: 26S proteasome regulatory subunit 8
+分子 #16: 26S protease regulatory subunit 6B
+分子 #17: 26S proteasome regulatory subunit 10B
+分子 #18: 26S protease regulatory subunit 6A
+分子 #19: Proteasome subunit alpha type-6
+分子 #20: Proteasome subunit alpha type-2
+分子 #21: Proteasome subunit alpha type-4
+分子 #22: Proteasome subunit alpha type-7
+分子 #23: Proteasome subunit alpha type-5
+分子 #24: Isoform Long of Proteasome subunit alpha type-1
+分子 #25: Proteasome subunit alpha type-3
+分子 #26: Proteasome subunit beta type-6
+分子 #27: Proteasome subunit beta type-7
+分子 #28: Proteasome subunit beta type-3
+分子 #29: Proteasome subunit beta type-2
+分子 #30: Proteasome subunit beta type-5
+分子 #31: Proteasome subunit beta type-1
+分子 #32: Proteasome subunit beta type-4
+分子 #33: ZINC ION
+分子 #34: ADENOSINE-5'-TRIPHOSPHATE
+分子 #35: MAGNESIUM ION
+分子 #36: ADENOSINE-5'-DIPHOSPHATE
-実験情報
-構造解析
| 手法 |  クライオ電子顕微鏡法 クライオ電子顕微鏡法 |
|---|---|
 解析 解析 |  単粒子再構成法 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 2 mg/mL |
|---|---|
| 緩衝液 | pH: 7 |
| 凍結 | 凍結剤: ETHANE |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | 照射モード: FLOOD BEAM / 撮影モード: BRIGHT FIELD Bright-field microscopy / 最大 デフォーカス(公称値): 2.5 µm / 最小 デフォーカス(公称値): 0.8 µm Bright-field microscopy / 最大 デフォーカス(公称値): 2.5 µm / 最小 デフォーカス(公称値): 0.8 µm |
| 撮影 | フィルム・検出器のモデル: GATAN K2 QUANTUM (4k x 4k) 平均電子線量: 46.6 e/Å2 |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 初期 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
|---|---|
| 最終 角度割当 | タイプ: MAXIMUM LIKELIHOOD |
| 最終 再構成 | 解像度のタイプ: BY AUTHOR / 解像度: 4.4 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 使用した粒子像数: 28928 |
 ムービー
ムービー コントローラー
コントローラー






















 Z (Sec.)
Z (Sec.) Y (Row.)
Y (Row.) X (Col.)
X (Col.)