+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 8uxx | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Arp2/3 branch junction complex, BeFx state | ||||||||||||
 Components Components |
| ||||||||||||
 Keywords Keywords |  CYTOSOLIC PROTEIN / CYTOSOLIC PROTEIN /  actin / actin /  arp2/3 / arp2/3 /  cytoskeleton / branch cytoskeleton / branch | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationRegulation of actin dynamics for phagocytic cup formation / RHO GTPases Activate WASPs and WAVEs / actin cortical patch organization / cell cortex of cell tip / medial cortex / Neutrophil degranulation / establishment or maintenance of cell polarity regulating cell shape / actin cortical patch assembly /  Arp2/3 protein complex / Arp2/3 complex-mediated actin nucleation ...Regulation of actin dynamics for phagocytic cup formation / RHO GTPases Activate WASPs and WAVEs / actin cortical patch organization / cell cortex of cell tip / medial cortex / Neutrophil degranulation / establishment or maintenance of cell polarity regulating cell shape / actin cortical patch assembly / Arp2/3 protein complex / Arp2/3 complex-mediated actin nucleation ...Regulation of actin dynamics for phagocytic cup formation / RHO GTPases Activate WASPs and WAVEs / actin cortical patch organization / cell cortex of cell tip / medial cortex / Neutrophil degranulation / establishment or maintenance of cell polarity regulating cell shape / actin cortical patch assembly /  Arp2/3 protein complex / Arp2/3 complex-mediated actin nucleation / cell tip / actin cortical patch / regulation of actin filament polymerization / mating projection tip / cortical actin cytoskeleton organization / cytoskeletal motor activator activity / cell division site / Arp2/3 protein complex / Arp2/3 complex-mediated actin nucleation / cell tip / actin cortical patch / regulation of actin filament polymerization / mating projection tip / cortical actin cytoskeleton organization / cytoskeletal motor activator activity / cell division site /  tropomyosin binding / tropomyosin binding /  myosin heavy chain binding / mesenchyme migration / myosin heavy chain binding / mesenchyme migration /  troponin I binding / actin filament bundle / filamentous actin / actin filament bundle assembly / skeletal muscle thin filament assembly / striated muscle thin filament / mitotic cytokinesis / skeletal muscle myofibril / actin monomer binding / skeletal muscle fiber development / troponin I binding / actin filament bundle / filamentous actin / actin filament bundle assembly / skeletal muscle thin filament assembly / striated muscle thin filament / mitotic cytokinesis / skeletal muscle myofibril / actin monomer binding / skeletal muscle fiber development /  stress fiber / stress fiber /  titin binding / actin filament polymerization / titin binding / actin filament polymerization /  filopodium / filopodium /  actin filament / actin filament /  Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement / structural constituent of cytoskeleton / Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement / structural constituent of cytoskeleton /  endocytosis / calcium-dependent protein binding / endocytosis / calcium-dependent protein binding /  actin filament binding / actin filament binding /  lamellipodium / lamellipodium /  cell body / cell body /  hydrolase activity / protein domain specific binding / hydrolase activity / protein domain specific binding /  calcium ion binding / positive regulation of gene expression / magnesium ion binding / calcium ion binding / positive regulation of gene expression / magnesium ion binding /  ATP binding / identical protein binding / ATP binding / identical protein binding /  nucleus / nucleus /  cytosol / cytosol /  cytoplasm cytoplasmSimilarity search - Function | ||||||||||||
| Biological species |   Schizosaccharomyces pombe (fission yeast) Schizosaccharomyces pombe (fission yeast)  Oryctolagus cuniculus (rabbit) Oryctolagus cuniculus (rabbit) | ||||||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.2 Å cryo EM / Resolution: 3.2 Å | ||||||||||||
 Authors Authors | Chavali, S.S. / Chou, S.Z. / Sindelar, C.V. | ||||||||||||
| Funding support |  United States, 3items United States, 3items
| ||||||||||||
 Citation Citation |  Journal: Nat Commun / Year: 2024 Journal: Nat Commun / Year: 2024Title: Cryo-EM structures reveal how phosphate release from Arp3 weakens actin filament branches formed by Arp2/3 complex. Authors: Sai Shashank Chavali / Steven Z Chou / Wenxiang Cao / Thomas D Pollard / Enrique M De La Cruz / Charles V Sindelar /  Abstract: Arp2/3 complex nucleates branched actin filaments for cell and organelle movements. Here we report a 2.7 Å resolution cryo-EM structure of the mature branch junction formed by S. pombe Arp2/3 ...Arp2/3 complex nucleates branched actin filaments for cell and organelle movements. Here we report a 2.7 Å resolution cryo-EM structure of the mature branch junction formed by S. pombe Arp2/3 complex that provides details about interactions with both mother and daughter filaments. We determine a second structure at 3.2 Å resolution with the phosphate analog BeF bound with ADP to Arp3 and ATP bound to Arp2. In this ADP-BeF transition state the outer domain of Arp3 is rotated 2° toward the mother filament compared with the ADP state and makes slightly broader contacts with actin in both the mother and daughter filaments. Thus, dissociation of P from the ADP-P transition state reduces the interactions of Arp2/3 complex with the actin filaments and may contribute to the lower mechanical stability of mature branch junctions with ADP bound to the Arps. Our structures also reveal that the mother filament in contact with Arp2/3 complex is slightly bent and twisted, consistent with the preference of Arp2/3 complex binding curved actin filaments. The small degree of twisting constrains models of actin filament mechanics. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  8uxx.cif.gz 8uxx.cif.gz | 1.6 MB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb8uxx.ent.gz pdb8uxx.ent.gz | 1.3 MB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  8uxx.json.gz 8uxx.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/ux/8uxx https://data.pdbj.org/pub/pdb/validation_reports/ux/8uxx ftp://data.pdbj.org/pub/pdb/validation_reports/ux/8uxx ftp://data.pdbj.org/pub/pdb/validation_reports/ux/8uxx | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  42788MC  8uxwC  8uz0C  8uz1C M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Actin-related protein ... , 7 types, 7 molecules ABCDEFG
| #1: Protein | Mass: 47427.137 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Schizosaccharomyces pombe (fission yeast) / References: UniProt: P32390 Schizosaccharomyces pombe (fission yeast) / References: UniProt: P32390 |
|---|---|
| #2: Protein | Mass: 44286.758 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Schizosaccharomyces pombe (fission yeast) / References: UniProt: Q9UUJ1 Schizosaccharomyces pombe (fission yeast) / References: UniProt: Q9UUJ1 |
| #3: Protein | Mass: 41643.465 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Schizosaccharomyces pombe (fission yeast) / References: UniProt: P78774 Schizosaccharomyces pombe (fission yeast) / References: UniProt: P78774 |
| #4: Protein | Mass: 37025.230 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Schizosaccharomyces pombe (fission yeast) / References: UniProt: O14241 Schizosaccharomyces pombe (fission yeast) / References: UniProt: O14241 |
| #5: Protein | Mass: 19865.746 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Schizosaccharomyces pombe (fission yeast) / References: UniProt: Q9Y7J4 Schizosaccharomyces pombe (fission yeast) / References: UniProt: Q9Y7J4 |
| #6: Protein | Mass: 19637.695 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Schizosaccharomyces pombe (fission yeast) / References: UniProt: Q92352 Schizosaccharomyces pombe (fission yeast) / References: UniProt: Q92352 |
| #7: Protein | Mass: 16922.059 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Schizosaccharomyces pombe (fission yeast) / References: UniProt: Q10316 Schizosaccharomyces pombe (fission yeast) / References: UniProt: Q10316 |
-Protein , 1 types, 8 molecules HIMNOPQR
| #8: Protein |  / Alpha-actin-1 / Alpha-actin-1Mass: 42109.973 Da / Num. of mol.: 8 / Source method: isolated from a natural source / Source: (natural)   Oryctolagus cuniculus (rabbit) Oryctolagus cuniculus (rabbit)References: UniProt: P68135,  Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement |
|---|
-Non-polymers , 5 types, 32 molecules 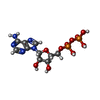

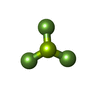
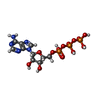


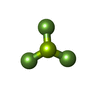


| #9: Chemical | ChemComp-ADP /  Adenosine diphosphate Adenosine diphosphate#10: Chemical | ChemComp-MG / #11: Chemical | ChemComp-BEF / #12: Chemical | ChemComp-ATP / |  Adenosine triphosphate Adenosine triphosphate#13: Water | ChemComp-HOH / |  Water Water |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| ||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Source (natural) |
| ||||||||||||||||||||||||||||
| Buffer solution | pH: 7.5 | ||||||||||||||||||||||||||||
| Specimen | Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES : YESDetails: Actin monomers with bound Ca2+ were converted to Mg2+-actin by equilibrating with 50 micromolar MgCl2 and 0.2 mM EGTA (pH 7.5) for 10 min on ice. Actin was polymerized in the presence of ...Details: Actin monomers with bound Ca2+ were converted to Mg2+-actin by equilibrating with 50 micromolar MgCl2 and 0.2 mM EGTA (pH 7.5) for 10 min on ice. Actin was polymerized in the presence of capping protein (CP) by sequentially mixing 8.75 micromolar Mg-ATP-actin monomers with 0.75 micromolar CP and equilibrated at room temperature for 1 h. In parallel, Arp2/3 complex (0.4 micromolar) in QB buffer was activated by mixing 0.85 micromolar GCN4-VCA and 50 micromolar ATP and incubated at 4 degrees for 1 h. The capped actin filaments sample was then gently mixed with an equal volume of activated Arp2/3 complex sample using cut pipette tips and equilibrated at 4 degrees for 5 min. Daughter filaments were formed and elongated in the presence of CP by adding 0.25 micromolar Mg-actin monomers, 50 micromolar ATP, and 40 nM CP and incubated for 5 min at room temperature. This step was subsequently repeated 4 more times, then aged for ~90 min before preparing grids for cryo-EM data collection. A final concentration of 2 mM BeFx (prepared using 2 mM BeSO4 and 10 mM NaF) was added during the 4th step of daughter filament formation (as mentioned above). | ||||||||||||||||||||||||||||
| Specimen support | Details: Grids were not glow discharged / Grid material: GOLD / Grid mesh size: 300 divisions/in. / Grid type: Quantifoil R1.2/1.3 | ||||||||||||||||||||||||||||
Vitrification | Instrument: FEI VITROBOT MARK IV / Cryogen name: ETHANE / Humidity: 100 % / Chamber temperature: 277 K Details: The samples were incubated on the grid for 50 s and the extra solution was blotted using two Vitrobot filter papers (0.55/20 mm, Grade 595, Ted Pella) for 4 s at 0 blot force. The grids were ...Details: The samples were incubated on the grid for 50 s and the extra solution was blotted using two Vitrobot filter papers (0.55/20 mm, Grade 595, Ted Pella) for 4 s at 0 blot force. The grids were plunged into liquid ethane at ~180 degrees C with a wait time of 0.5 s. |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS Details: The vitrified grids were screened for sample homogeneity and ice thickness in a Glacios 200 kV transmission electron microscope equipped with Gatan K2 summit camera. Electron micrographs for ...Details: The vitrified grids were screened for sample homogeneity and ice thickness in a Glacios 200 kV transmission electron microscope equipped with Gatan K2 summit camera. Electron micrographs for image reconstructions were collected using Titan Krios equipped with X-cold field emission gun at 300 kV, Gatan image filter with slit width of 20 eV and a nanoprobe. |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: FLOOD BEAM |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal magnification: 64000 X / Nominal defocus max: 2500 nm / Nominal defocus min: 1200 nm / Cs Bright-field microscopy / Nominal magnification: 64000 X / Nominal defocus max: 2500 nm / Nominal defocus min: 1200 nm / Cs : 2.7 mm : 2.7 mm |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 3.3 sec. / Electron dose: 51.4 e/Å2 / Film or detector model: GATAN K3 (6k x 4k) / Num. of real images: 8519 Details: Each movie contains 41 frames with a frame time of 0.08 s. A dose rate of 28.4 counts/pixel/s and a physical pixel size of 1.346 Angstroms was used. |
| EM imaging optics | Energyfilter name : GIF Quantum LS : GIF Quantum LSDetails: Electron micrographs for image reconstructions were collected using Titan Krios equipped with X-cold field emission gun at 300 kV, Gatan image filter with slit width of 20 eV and a nanoprobe. Energyfilter slit width: 20 eV |
- Processing
Processing
| EM software |
| ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
CTF correction | Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | ||||||||||||||||||||
| Particle selection | Num. of particles selected: 3000000 | ||||||||||||||||||||
3D reconstruction | Resolution: 3.2 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 200000 / Symmetry type: POINT |
 Movie
Movie Controller
Controller






 PDBj
PDBj










