+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 | データベース: PDB / ID: 8sc6 | ||||||
|---|---|---|---|---|---|---|---|
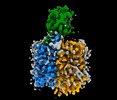
| タイトル | Human OCT1 bound to thiamine in inward-open conformation | ||||||
 要素 要素 | Solute carrier family 22 member 1 | ||||||
 キーワード キーワード |  TRANSPORT PROTEIN (運搬体タンパク質) / TRANSPORT PROTEIN (運搬体タンパク質) /  membrane protein (膜タンパク質) / MFS / drug transporter membrane protein (膜タンパク質) / MFS / drug transporter | ||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報acetylcholine transmembrane transporter activity / secondary active organic cation transmembrane transporter activity / putrescine transmembrane transporter activity / (R)-carnitine transmembrane transporter activity / acyl carnitine transmembrane transport / pyrimidine nucleoside transmembrane transporter activity / acetylcholine transport / serotonin transport / spermidine transmembrane transporter activity / quaternary ammonium group transmembrane transporter activity ...acetylcholine transmembrane transporter activity / secondary active organic cation transmembrane transporter activity / putrescine transmembrane transporter activity / (R)-carnitine transmembrane transporter activity / acyl carnitine transmembrane transport / pyrimidine nucleoside transmembrane transporter activity / acetylcholine transport / serotonin transport / spermidine transmembrane transporter activity / quaternary ammonium group transmembrane transporter activity / quaternary ammonium group transport / epinephrine transport / thiamine transmembrane transport / purine-containing compound transmembrane transport / spermidine transport / thiamine transport / thiamine transmembrane transporter activity /  organic cation transport / organic cation transport /  Organic cation transport / organic cation transmembrane transporter activity / putrescine transport / dopamine uptake / metanephric proximal tubule development / : / dopamine:sodium symporter activity / norepinephrine:sodium symporter activity / prostaglandin transport / norepinephrine transport / prostaglandin transmembrane transporter activity / Norepinephrine Neurotransmitter Release Cycle / toxin transmembrane transporter activity / neurotransmitter transmembrane transporter activity / serotonin uptake / Abacavir transmembrane transport / dopamine transport / Na+/Cl- dependent neurotransmitter transporters / monoamine transmembrane transporter activity / monoamine transport / organic anion transmembrane transporter activity / establishment or maintenance of transmembrane electrochemical gradient / xenobiotic transport across blood-brain barrier / Neurotransmitter clearance / Ciprofloxacin ADME / xenobiotic transport / Organic cation transport / organic cation transmembrane transporter activity / putrescine transport / dopamine uptake / metanephric proximal tubule development / : / dopamine:sodium symporter activity / norepinephrine:sodium symporter activity / prostaglandin transport / norepinephrine transport / prostaglandin transmembrane transporter activity / Norepinephrine Neurotransmitter Release Cycle / toxin transmembrane transporter activity / neurotransmitter transmembrane transporter activity / serotonin uptake / Abacavir transmembrane transport / dopamine transport / Na+/Cl- dependent neurotransmitter transporters / monoamine transmembrane transporter activity / monoamine transport / organic anion transmembrane transporter activity / establishment or maintenance of transmembrane electrochemical gradient / xenobiotic transport across blood-brain barrier / Neurotransmitter clearance / Ciprofloxacin ADME / xenobiotic transport /  神経伝達物質輸送体 / xenobiotic transmembrane transporter activity / transport across blood-brain barrier / lateral plasma membrane / xenobiotic metabolic process / basal plasma membrane / presynapse / basolateral plasma membrane / apical plasma membrane / 神経伝達物質輸送体 / xenobiotic transmembrane transporter activity / transport across blood-brain barrier / lateral plasma membrane / xenobiotic metabolic process / basal plasma membrane / presynapse / basolateral plasma membrane / apical plasma membrane /  生体膜 / identical protein binding / 生体膜 / identical protein binding /  細胞膜 細胞膜類似検索 - 分子機能 | ||||||
| 生物種 |   Homo sapiens (ヒト) Homo sapiens (ヒト) | ||||||
| 手法 |  電子顕微鏡法 / 電子顕微鏡法 /  単粒子再構成法 / 単粒子再構成法 /  クライオ電子顕微鏡法 / 解像度: 3.13 Å クライオ電子顕微鏡法 / 解像度: 3.13 Å | ||||||
 データ登録者 データ登録者 | Zeng, Y.C. / Sobti, M. / Stewart, A.G. | ||||||
| 資金援助 |  オーストラリア, 1件 オーストラリア, 1件
| ||||||
 引用 引用 |  ジャーナル: Nat Commun / 年: 2023 ジャーナル: Nat Commun / 年: 2023タイトル: Structural basis of promiscuous substrate transport by Organic Cation Transporter 1. 著者: Yi C Zeng / Meghna Sobti / Ada Quinn / Nicola J Smith / Simon H J Brown / Jamie I Vandenberg / Renae M Ryan / Megan L O'Mara / Alastair G Stewart /  要旨: Organic Cation Transporter 1 (OCT1) plays a crucial role in hepatic metabolism by mediating the uptake of a range of metabolites and drugs. Genetic variations can alter the efficacy and safety of ...Organic Cation Transporter 1 (OCT1) plays a crucial role in hepatic metabolism by mediating the uptake of a range of metabolites and drugs. Genetic variations can alter the efficacy and safety of compounds transported by OCT1, such as those used for cardiovascular, oncological, and psychological indications. Despite its importance in drug pharmacokinetics, the substrate selectivity and underlying structural mechanisms of OCT1 remain poorly understood. Here, we present cryo-EM structures of full-length human OCT1 in the inward-open conformation, both ligand-free and drug-bound, indicating the basis for its broad substrate recognition. Comparison of our structures with those of outward-open OCTs provides molecular insight into the alternating access mechanism of OCTs. We observe that hydrophobic gates stabilize the inward-facing conformation, whereas charge neutralization in the binding pocket facilitates the release of cationic substrates. These findings provide a framework for understanding the structural basis of the promiscuity of drug binding and substrate translocation in OCT1. | ||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 構造ビューア | 分子:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- ダウンロードとリンク
ダウンロードとリンク
- ダウンロード
ダウンロード
| PDBx/mmCIF形式 |  8sc6.cif.gz 8sc6.cif.gz | 99.9 KB | 表示 |  PDBx/mmCIF形式 PDBx/mmCIF形式 |
|---|---|---|---|---|
| PDB形式 |  pdb8sc6.ent.gz pdb8sc6.ent.gz | 73.3 KB | 表示 |  PDB形式 PDB形式 |
| PDBx/mmJSON形式 |  8sc6.json.gz 8sc6.json.gz | ツリー表示 |  PDBx/mmJSON形式 PDBx/mmJSON形式 | |
| その他 |  その他のダウンロード その他のダウンロード |
-検証レポート
| アーカイブディレクトリ |  https://data.pdbj.org/pub/pdb/validation_reports/sc/8sc6 https://data.pdbj.org/pub/pdb/validation_reports/sc/8sc6 ftp://data.pdbj.org/pub/pdb/validation_reports/sc/8sc6 ftp://data.pdbj.org/pub/pdb/validation_reports/sc/8sc6 | HTTPS FTP |
|---|
-関連構造データ
| 関連構造データ |  40339MC  8sc1C  8sc2C  8sc3C  8sc4C M: このデータのモデリングに利用したマップデータ C: 同じ文献を引用 ( |
|---|---|
| 類似構造データ | 類似検索 - 機能・相同性  F&H 検索 F&H 検索 |
- リンク
リンク
- 集合体
集合体
| 登録構造単位 | 
|
|---|---|
| 1 |
|
- 要素
要素
| #1: タンパク質 | 分子量: 61200.664 Da / 分子数: 1 / 由来タイプ: 組換発現 / 由来: (組換発現)   Homo sapiens (ヒト) / 遺伝子: SLC22A1, OCT1 / 発現宿主: Homo sapiens (ヒト) / 遺伝子: SLC22A1, OCT1 / 発現宿主:   Homo sapiens (ヒト) / 参照: UniProt: O15245 Homo sapiens (ヒト) / 参照: UniProt: O15245 |
|---|---|
| #2: 化合物 | ChemComp-VIB /  チアミン チアミン |
| 研究の焦点であるリガンドがあるか | Y |
-実験情報
-実験
| 実験 | 手法:  電子顕微鏡法 電子顕微鏡法 |
|---|---|
| EM実験 | 試料の集合状態: PARTICLE / 3次元再構成法:  単粒子再構成法 単粒子再構成法 |
- 試料調製
試料調製
| 構成要素 | 名称: Human OCT1 / タイプ: COMPLEX / Entity ID: #1 / 由来: RECOMBINANT |
|---|---|
| 由来(天然) | 生物種:   Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 由来(組換発現) | 生物種:   Homo sapiens (ヒト) Homo sapiens (ヒト) |
| 緩衝液 | pH: 8 |
| 試料 | 包埋: NO / シャドウイング: NO / 染色 : NO / 凍結 : NO / 凍結 : YES : YES |
急速凍結 | 凍結剤: ETHANE |
- 電子顕微鏡撮影
電子顕微鏡撮影
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
|---|---|
| 顕微鏡 | モデル: FEI TITAN KRIOS |
| 電子銃 | 電子線源 : :  FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM FIELD EMISSION GUN / 加速電圧: 300 kV / 照射モード: FLOOD BEAM |
| 電子レンズ | モード: BRIGHT FIELD Bright-field microscopy / 最大 デフォーカス(公称値): 2000 nm / 最小 デフォーカス(公称値): 500 nm Bright-field microscopy / 最大 デフォーカス(公称値): 2000 nm / 最小 デフォーカス(公称値): 500 nm |
| 撮影 | 電子線照射量: 88 e/Å2 / フィルム・検出器のモデル: GATAN K3 (6k x 4k) |
- 解析
解析
CTF補正 | タイプ: PHASE FLIPPING ONLY |
|---|---|
3次元再構成 | 解像度: 3.13 Å / 解像度の算出法: FSC 0.143 CUT-OFF / 粒子像の数: 1105286 / 対称性のタイプ: POINT |
 ムービー
ムービー コントローラー
コントローラー






 PDBj
PDBj