[English] 日本語
 Yorodumi
Yorodumi- PDB-7t5q: Cryo-EM Structure of a Transition State of Arp2/3 Complex Activation -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7t5q | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Title | Cryo-EM Structure of a Transition State of Arp2/3 Complex Activation | ||||||||||||
 Components Components |
| ||||||||||||
 Keywords Keywords |  CONTRACTILE PROTEIN / CONTRACTILE PROTEIN /  actin / actin /  ATPase / actin related protein / arp / ATPase / actin related protein / arp /  cytoskeleton / Arp2-3 complex / actin nucleation / actin branching / cytoskeleton / Arp2-3 complex / actin nucleation / actin branching /  CapZ CapZ | ||||||||||||
| Function / homology |  Function and homology information Function and homology informationnegative regulation of membrane tubulation / muscle cell projection membrane / spindle localization / membrane invagination / positive regulation of clathrin-dependent endocytosis / EPHB-mediated forward signaling / Regulation of actin dynamics for phagocytic cup formation / RHO GTPases Activate WASPs and WAVEs / plasma membrane tubulation /  Arp2/3 protein complex ...negative regulation of membrane tubulation / muscle cell projection membrane / spindle localization / membrane invagination / positive regulation of clathrin-dependent endocytosis / EPHB-mediated forward signaling / Regulation of actin dynamics for phagocytic cup formation / RHO GTPases Activate WASPs and WAVEs / plasma membrane tubulation / Arp2/3 protein complex ...negative regulation of membrane tubulation / muscle cell projection membrane / spindle localization / membrane invagination / positive regulation of clathrin-dependent endocytosis / EPHB-mediated forward signaling / Regulation of actin dynamics for phagocytic cup formation / RHO GTPases Activate WASPs and WAVEs / plasma membrane tubulation /  Arp2/3 protein complex / negative regulation of lymphocyte migration / Arp2/3 complex-mediated actin nucleation / WASH complex / postsynapse organization / sperm connecting piece / actin nucleation / F-actin capping protein complex / negative regulation of filopodium assembly / regulation of cell projection assembly / actin cap / vesicle organization / postsynaptic actin cytoskeleton organization / vesicle budding from membrane / regulation of actin filament polymerization / positive regulation of chemotaxis / Arp2/3 protein complex / negative regulation of lymphocyte migration / Arp2/3 complex-mediated actin nucleation / WASH complex / postsynapse organization / sperm connecting piece / actin nucleation / F-actin capping protein complex / negative regulation of filopodium assembly / regulation of cell projection assembly / actin cap / vesicle organization / postsynaptic actin cytoskeleton organization / vesicle budding from membrane / regulation of actin filament polymerization / positive regulation of chemotaxis /  Clathrin-mediated endocytosis / Clathrin-mediated endocytosis /  cell junction assembly / COPI-independent Golgi-to-ER retrograde traffic / barbed-end actin filament capping / vesicle transport along actin filament / actin polymerization or depolymerization / protein-containing complex localization / dendritic spine morphogenesis / RHOF GTPase cycle / RHOD GTPase cycle / regulation of cell morphogenesis / cell junction assembly / COPI-independent Golgi-to-ER retrograde traffic / barbed-end actin filament capping / vesicle transport along actin filament / actin polymerization or depolymerization / protein-containing complex localization / dendritic spine morphogenesis / RHOF GTPase cycle / RHOD GTPase cycle / regulation of cell morphogenesis /  regulation of lamellipodium assembly / Sensory processing of sound by inner hair cells of the cochlea / Neutrophil degranulation / regulation of postsynapse organization / regulation of lamellipodium assembly / Sensory processing of sound by inner hair cells of the cochlea / Neutrophil degranulation / regulation of postsynapse organization /  lamellipodium assembly / positive regulation of filopodium assembly / cytoskeletal motor activator activity / lamellipodium assembly / positive regulation of filopodium assembly / cytoskeletal motor activator activity /  tropomyosin binding / positive regulation of double-strand break repair via homologous recombination / tropomyosin binding / positive regulation of double-strand break repair via homologous recombination /  myosin heavy chain binding / cortical cytoskeleton / mesenchyme migration / myosin heavy chain binding / cortical cytoskeleton / mesenchyme migration /  troponin I binding / cell leading edge / actin filament bundle / filamentous actin / actin filament bundle assembly / troponin I binding / cell leading edge / actin filament bundle / filamentous actin / actin filament bundle assembly /  brush border / skeletal muscle thin filament assembly / Advanced glycosylation endproduct receptor signaling / striated muscle thin filament / brush border / skeletal muscle thin filament assembly / Advanced glycosylation endproduct receptor signaling / striated muscle thin filament /  cilium assembly / skeletal muscle myofibril / actin monomer binding / positive regulation of lamellipodium assembly / COPI-mediated anterograde transport / skeletal muscle fiber development / cilium assembly / skeletal muscle myofibril / actin monomer binding / positive regulation of lamellipodium assembly / COPI-mediated anterograde transport / skeletal muscle fiber development /  stress fiber / stress fiber /  titin binding / cytoskeleton organization / HSP90 chaperone cycle for steroid hormone receptors (SHR) in the presence of ligand / hippocampal mossy fiber to CA3 synapse / MHC class II antigen presentation / actin filament polymerization / Gene and protein expression by JAK-STAT signaling after Interleukin-12 stimulation / titin binding / cytoskeleton organization / HSP90 chaperone cycle for steroid hormone receptors (SHR) in the presence of ligand / hippocampal mossy fiber to CA3 synapse / MHC class II antigen presentation / actin filament polymerization / Gene and protein expression by JAK-STAT signaling after Interleukin-12 stimulation /  cytoskeletal protein binding / cytoskeletal protein binding /  sarcomere / sarcomere /  filopodium / cell projection / filopodium / cell projection /  actin filament / response to bacterium / actin filament / response to bacterium /  Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement / Schaffer collateral - CA1 synapse / cell morphogenesis / structural constituent of cytoskeleton / calcium-dependent protein binding / Hydrolases; Acting on acid anhydrides; Acting on acid anhydrides to facilitate cellular and subcellular movement / Schaffer collateral - CA1 synapse / cell morphogenesis / structural constituent of cytoskeleton / calcium-dependent protein binding /  regulation of protein localization / regulation of protein localization /  cell migration / cell migration /  actin filament binding / actin filament binding /  actin cytoskeleton / actin cytoskeleton /  lamellipodium / site of double-strand break / lamellipodium / site of double-strand break /  cell body / cell body /  actin binding / Factors involved in megakaryocyte development and platelet production / cytoplasmic vesicle / actin cytoskeleton organization / postsynapse / protein-containing complex assembly / actin binding / Factors involved in megakaryocyte development and platelet production / cytoplasmic vesicle / actin cytoskeleton organization / postsynapse / protein-containing complex assembly /  postsynaptic density / postsynaptic density /  cytoskeleton / cytoskeleton /  hydrolase activity / neuron projection / hydrolase activity / neuron projection /  cadherin binding cadherin bindingSimilarity search - Function | ||||||||||||
| Biological species |   Homo sapiens (human) Homo sapiens (human)  Mus musculus (house mouse) Mus musculus (house mouse)  Bos taurus (cattle) Bos taurus (cattle)  Oryctolagus cuniculus (rabbit) Oryctolagus cuniculus (rabbit) | ||||||||||||
| Method |  ELECTRON MICROSCOPY / ELECTRON MICROSCOPY /  single particle reconstruction / single particle reconstruction /  cryo EM / Resolution: 3.4 Å cryo EM / Resolution: 3.4 Å | ||||||||||||
 Authors Authors | Rebowski, G. / van Eeuwen, T. / Boczkowska, M. / Dominguez, R. | ||||||||||||
| Funding support |  United States, 3items United States, 3items
| ||||||||||||
 Citation Citation |  Journal: To Be Published Journal: To Be PublishedTitle: Cryo-EM structure of an Arp2/3 complex mini-branch capped by capping protein (CapZ) Authors: Rebowski, G. / van Eeuwen, T. / Dominguez, R. / Boczkowska, M. | ||||||||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7t5q.cif.gz 7t5q.cif.gz | 547.8 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7t5q.ent.gz pdb7t5q.ent.gz | 438.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  7t5q.json.gz 7t5q.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/t5/7t5q https://data.pdbj.org/pub/pdb/validation_reports/t5/7t5q ftp://data.pdbj.org/pub/pdb/validation_reports/t5/7t5q ftp://data.pdbj.org/pub/pdb/validation_reports/t5/7t5q | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  25707MC M: map data used to model this data C: citing same article ( |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
|
|---|---|
| 1 |
|
- Components
Components
-Actin-related protein ... , 7 types, 7 molecules ABCDEFG
| #1: Protein | Mass: 47428.031 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Bos taurus (cattle) / References: UniProt: P61157 Bos taurus (cattle) / References: UniProt: P61157 |
|---|---|
| #2: Protein | Mass: 44818.711 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Bos taurus (cattle) / References: UniProt: A7MB62 Bos taurus (cattle) / References: UniProt: A7MB62 |
| #3: Protein | Mass: 41594.238 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Bos taurus (cattle) / References: UniProt: Q1JP79 Bos taurus (cattle) / References: UniProt: Q1JP79 |
| #4: Protein | Mass: 34402.043 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Bos taurus (cattle) / References: UniProt: Q3MHR7 Bos taurus (cattle) / References: UniProt: Q3MHR7 |
| #5: Protein | Mass: 20572.666 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Bos taurus (cattle) / References: UniProt: Q3T035 Bos taurus (cattle) / References: UniProt: Q3T035 |
| #6: Protein | Mass: 19697.047 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Bos taurus (cattle) / References: UniProt: Q148J6 Bos taurus (cattle) / References: UniProt: Q148J6 |
| #7: Protein | Mass: 16251.308 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Bos taurus (cattle) / References: UniProt: Q3SYX9 Bos taurus (cattle) / References: UniProt: Q3SYX9 |
-Protein , 1 types, 1 molecules H
| #8: Protein |  / Alpha-actin-1 / Alpha-actin-1Mass: 41875.633 Da / Num. of mol.: 1 / Source method: isolated from a natural source / Source: (natural)   Oryctolagus cuniculus (rabbit) / References: UniProt: P68135 Oryctolagus cuniculus (rabbit) / References: UniProt: P68135 |
|---|
-F-actin-capping protein subunit ... , 2 types, 2 molecules IJ
| #9: Protein | Mass: 39600.566 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human), (gene. exp.) Homo sapiens (human), (gene. exp.)   Mus musculus (house mouse) Mus musculus (house mouse)Gene: CAPZA1, Wasl / Production host:   Escherichia coli (E. coli) / References: UniProt: P52907, UniProt: Q91YD9 Escherichia coli (E. coli) / References: UniProt: P52907, UniProt: Q91YD9 |
|---|---|
| #10: Protein | Mass: 37470.754 Da / Num. of mol.: 1 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Homo sapiens (human), (gene. exp.) Homo sapiens (human), (gene. exp.)   Mus musculus (house mouse) Mus musculus (house mouse)Gene: CAPZB, Wasl / Production host:   Escherichia coli (E. coli) / References: UniProt: P47756, UniProt: Q91YD9 Escherichia coli (E. coli) / References: UniProt: P47756, UniProt: Q91YD9 |
-Non-polymers , 3 types, 6 molecules 
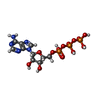
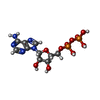


| #11: Chemical | | #12: Chemical |  Adenosine triphosphate Adenosine triphosphate#13: Chemical | ChemComp-ADP / |  Adenosine diphosphate Adenosine diphosphate |
|---|
-Details
| Has ligand of interest | N |
|---|
-Experimental details
-Experiment
| Experiment | Method:  ELECTRON MICROSCOPY ELECTRON MICROSCOPY |
|---|---|
| EM experiment | Aggregation state: PARTICLE / 3D reconstruction method:  single particle reconstruction single particle reconstruction |
- Sample preparation
Sample preparation
| Component |
| |||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Molecular weight | Value: 0.2489 MDa / Experimental value: NO | |||||||||||||||||||||||||||||||||||
| Source (natural) |
| |||||||||||||||||||||||||||||||||||
| Source (recombinant) |
| |||||||||||||||||||||||||||||||||||
| Buffer solution | pH: 7 | |||||||||||||||||||||||||||||||||||
| Buffer component |
| |||||||||||||||||||||||||||||||||||
| Specimen | Conc.: 5 mg/ml / Embedding applied: NO / Shadowing applied: NO / Staining applied : NO / Vitrification applied : NO / Vitrification applied : YES / Details: This sample was monodisperse. : YES / Details: This sample was monodisperse. | |||||||||||||||||||||||||||||||||||
Vitrification | Instrument: LEICA EM CPC / Cryogen name: ETHANE Details: Grids were manually blotted for 3 seconds with Whatman 41 filter paper and manually plunged using a Leica EM CPC manual plunger. |
- Electron microscopy imaging
Electron microscopy imaging
| Experimental equipment |  Model: Titan Krios / Image courtesy: FEI Company |
|---|---|
| Microscopy | Model: FEI TITAN KRIOS Details: Data were collected in super-resolution mode with an illuminated area of 1.01 um, nominal dose of 40 e-/A^2, a dose rate of 4.87 e-/s/pixel, and 2 or 5 exposures per hole by image shift. |
| Electron gun | Electron source : :  FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN FIELD EMISSION GUN / Accelerating voltage: 300 kV / Illumination mode: SPOT SCAN |
| Electron lens | Mode: BRIGHT FIELD Bright-field microscopy / Nominal magnification: 165000 X / Nominal defocus max: 3500 nm / Nominal defocus min: 1500 nm / Cs Bright-field microscopy / Nominal magnification: 165000 X / Nominal defocus max: 3500 nm / Nominal defocus min: 1500 nm / Cs : 2.7 mm / C2 aperture diameter: 100 µm / Alignment procedure: COMA FREE : 2.7 mm / C2 aperture diameter: 100 µm / Alignment procedure: COMA FREE |
| Specimen holder | Cryogen: NITROGEN / Specimen holder model: FEI TITAN KRIOS AUTOGRID HOLDER |
| Image recording | Average exposure time: 2.6 sec. / Electron dose: 50 e/Å2 / Detector mode: SUPER-RESOLUTION / Film or detector model: GATAN K3 (6k x 4k) / Num. of grids imaged: 1 / Num. of real images: 9661 |
| EM imaging optics | Energyfilter slit width: 20 eV |
| Image scans | Width: 5760 / Height: 4092 / Movie frames/image: 40 / Used frames/image: 1-40 |
- Processing
Processing
| Software | Name: PHENIX / Version: 1.20.1_4487: / Classification: refinement | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| EM software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Image processing | Details: Super resolution mode; micrographs binned during motion correction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
CTF correction | Details: CTF correction was done in cryoSPARC for intial 2D classification and then repeated in CTFFIND4 (Relion) for classification and final reconstruction. Type: PHASE FLIPPING AND AMPLITUDE CORRECTION | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Particle selection | Num. of particles selected: 4485066 / Details: Particles autopicked in cryoSPARC | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Symmetry | Point symmetry : C1 (asymmetric) : C1 (asymmetric) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
3D reconstruction | Resolution: 3.4 Å / Resolution method: FSC 0.143 CUT-OFF / Num. of particles: 269760 / Algorithm: FOURIER SPACE / Details: Composite map after multibody refinement / Num. of class averages: 1 / Symmetry type: POINT | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | B value: 132 / Protocol: FLEXIBLE FIT / Space: REAL / Target criteria: correlation | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic model building | PDB-ID: 4JD2 Accession code: 4JD2 / Source name: PDB / Type: experimental model | |||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller


 PDBj
PDBj





























