[English] 日本語
 Yorodumi
Yorodumi- PDB-7p9y: N-acetylglucosamine kinase from Plesiomonas shigelloides compexed... -
+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 7p9y | ||||||
|---|---|---|---|---|---|---|---|
| Title | N-acetylglucosamine kinase from Plesiomonas shigelloides compexed with alpha-N-acetylglucosamine | ||||||
 Components Components | Ubiquitin-like protein SMT3,N-acetyl-D-glucosamine kinase | ||||||
 Keywords Keywords | SUGAR BINDING PROTEIN / N-acetylglucosamine recycling / carbohydrate kinase / ROK kinase. | ||||||
| Function / homology |  Function and homology information Function and homology information N-acetylglucosamine kinase / N-acetylglucosamine kinase /  N-acetylglucosamine kinase activity / N-acetylglucosamine metabolic process / SUMO is conjugated to E1 (UBA2:SAE1) / SUMOylation of nuclear envelope proteins / SUMO is transferred from E1 to E2 (UBE2I, UBC9) / SUMOylation of SUMOylation proteins / SUMO is proteolytically processed / SUMOylation of transcription factors / Postmitotic nuclear pore complex (NPC) reformation ... N-acetylglucosamine kinase activity / N-acetylglucosamine metabolic process / SUMO is conjugated to E1 (UBA2:SAE1) / SUMOylation of nuclear envelope proteins / SUMO is transferred from E1 to E2 (UBE2I, UBC9) / SUMOylation of SUMOylation proteins / SUMO is proteolytically processed / SUMOylation of transcription factors / Postmitotic nuclear pore complex (NPC) reformation ... N-acetylglucosamine kinase / N-acetylglucosamine kinase /  N-acetylglucosamine kinase activity / N-acetylglucosamine metabolic process / SUMO is conjugated to E1 (UBA2:SAE1) / SUMOylation of nuclear envelope proteins / SUMO is transferred from E1 to E2 (UBE2I, UBC9) / SUMOylation of SUMOylation proteins / SUMO is proteolytically processed / SUMOylation of transcription factors / Postmitotic nuclear pore complex (NPC) reformation / SUMOylation of transcription cofactors / N-acetylglucosamine kinase activity / N-acetylglucosamine metabolic process / SUMO is conjugated to E1 (UBA2:SAE1) / SUMOylation of nuclear envelope proteins / SUMO is transferred from E1 to E2 (UBE2I, UBC9) / SUMOylation of SUMOylation proteins / SUMO is proteolytically processed / SUMOylation of transcription factors / Postmitotic nuclear pore complex (NPC) reformation / SUMOylation of transcription cofactors /  septin ring / SUMOylation of DNA damage response and repair proteins / SUMOylation of RNA binding proteins / peptidoglycan turnover / SUMOylation of DNA replication proteins / SUMOylation of chromatin organization proteins / ubiquitin-like protein ligase binding / protein sumoylation / condensed nuclear chromosome / septin ring / SUMOylation of DNA damage response and repair proteins / SUMOylation of RNA binding proteins / peptidoglycan turnover / SUMOylation of DNA replication proteins / SUMOylation of chromatin organization proteins / ubiquitin-like protein ligase binding / protein sumoylation / condensed nuclear chromosome /  protein tag activity / zinc ion binding / protein tag activity / zinc ion binding /  ATP binding / identical protein binding / ATP binding / identical protein binding /  nucleus nucleusSimilarity search - Function | ||||||
| Biological species |   Saccharomyces cerevisiae (brewer's yeast) Saccharomyces cerevisiae (brewer's yeast)  Plesiomonas shigelloides 302-73 (bacteria) Plesiomonas shigelloides 302-73 (bacteria) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.94 Å MOLECULAR REPLACEMENT / Resolution: 1.94 Å | ||||||
 Authors Authors | Roy, S. / Isupov, M.N. / Harmer, N.J. / Ames, J.R. | ||||||
| Funding support |  United Kingdom, 1items United Kingdom, 1items
| ||||||
 Citation Citation |  Journal: J.Biol.Chem. / Year: 2022 Journal: J.Biol.Chem. / Year: 2022Title: Spinning sugars in antigen biosynthesis: characterization of the Coxiella burnetii and Streptomyces griseus TDP-sugar epimerases Authors: Cross, A.R. / Roy, S. / Vivoli Vega, M. / Rejzek, M. / Nepogodiev, S.A. / Cliff, M. / Salmon, D. / Isupov, M.N. / Field, R.A. / Prior, J.L. / Harmer, N.J. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  7p9y.cif.gz 7p9y.cif.gz | 153.2 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb7p9y.ent.gz pdb7p9y.ent.gz | Display |  PDB format PDB format | |
| PDBx/mmJSON format |  7p9y.json.gz 7p9y.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/p9/7p9y https://data.pdbj.org/pub/pdb/validation_reports/p9/7p9y ftp://data.pdbj.org/pub/pdb/validation_reports/p9/7p9y ftp://data.pdbj.org/pub/pdb/validation_reports/p9/7p9y | HTTPS FTP |
|---|
-Related structure data
| Related structure data |  7p7iC  7p7wC  7p9lC  7p9pC  7pa1C  4db3S C: citing same article ( S: Starting model for refinement |
|---|---|
| Similar structure data | Similarity search - Function & homology  F&H Search F&H Search |
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| |||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| |||||||||||||||||||||
| Unit cell |
| |||||||||||||||||||||
| Noncrystallographic symmetry (NCS) | NCS domain:
NCS domain segments: Ens-ID: 1 / Beg auth comp-ID: GLY / Beg label comp-ID: GLY / End auth comp-ID: LEU / End label comp-ID: LEU / Auth seq-ID: -1 - 302 / Label seq-ID: 114 - 417
NCS ensembles : (Details: Local NCS retraints between domains: 1 2) |
- Components
Components
-Protein / Sugars , 2 types, 4 molecules AAABBB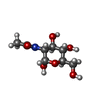

| #1: Protein | Mass: 45960.145 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Saccharomyces cerevisiae (strain ATCC 204508 / S288c) (yeast), (gene. exp.) Saccharomyces cerevisiae (strain ATCC 204508 / S288c) (yeast), (gene. exp.)   Plesiomonas shigelloides 302-73 (bacteria) Plesiomonas shigelloides 302-73 (bacteria)Strain: ATCC 204508 / S288c / Gene: SMT3, YDR510W, D9719.15, nagK, PLESHI_11010 / Production host:   Escherichia coli BL21(DE3) (bacteria) Escherichia coli BL21(DE3) (bacteria)References: UniProt: Q12306, UniProt: R8APY9,  N-acetylglucosamine kinase N-acetylglucosamine kinase#5: Sugar |  N-Acetylglucosamine N-Acetylglucosamine |
|---|
-Non-polymers , 8 types, 302 molecules 
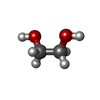
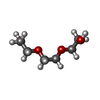












| #2: Chemical |  2-Methyl-2,4-pentanediol 2-Methyl-2,4-pentanediol#3: Chemical | ChemComp-EDO /  Ethylene glycol Ethylene glycol#4: Chemical | ChemComp-PGE / |  Polyethylene glycol Polyethylene glycol#6: Chemical | ChemComp-ZN / #7: Chemical | #8: Chemical |  Chloride Chloride#9: Chemical | ChemComp-PEG / |  Diethylene glycol Diethylene glycol#10: Water | ChemComp-HOH / |  Water Water |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.51 Å3/Da / Density % sol: 50.91 % |
|---|---|
Crystal grow | Temperature: 291 K / Method: microbatch Details: 90 mM halogens; 0.1 M Na-HEPES/MOPS pH 7.5; 12.5% each MPD, PEG 1K, PEG 3350 Condition B8 of Morpheus screen (Molecular Dimensions) |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  Diamond Diamond  / Beamline: I03 / Wavelength: 0.9763 Å / Beamline: I03 / Wavelength: 0.9763 Å |
| Detector | Type: DECTRIS PILATUS 6M / Detector: PIXEL / Date: Aug 5, 2018 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 0.9763 Å / Relative weight: 1 : 0.9763 Å / Relative weight: 1 |
| Reflection | Resolution: 1.94→57.74 Å / Num. obs: 68568 / % possible obs: 100 % / Redundancy: 6.7 % / CC1/2: 0.999 / Net I/σ(I): 11 |
| Reflection shell | Resolution: 1.94→1.99 Å / Num. unique obs: 4548 / CC1/2: 0.282 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: 4DB3 Resolution: 1.94→46.136 Å / Cor.coef. Fo:Fc: 0.962 / Cor.coef. Fo:Fc free: 0.944 / SU B: 4.603 / SU ML: 0.122 / Cross valid method: FREE R-VALUE / ESU R: 0.139 / ESU R Free: 0.138 / Details: Hydrogens have not been used
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.2 Å / Solvent model: BABINET MODEL PLUS MASK | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 55.854 Å2
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.94→46.136 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints NCS |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell | Refine-ID: X-RAY DIFFRACTION / Total num. of bins used: 20
|
 Movie
Movie Controller
Controller



 PDBj
PDBj












