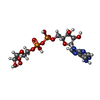
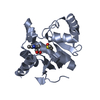
Entry Database : PDB / ID : 7kqpTitle Crystal structure of SARS-CoV-2 NSP3 macrodomain in complex with ADP-ribose (P43 crystal form) Non-structural protein 3 Keywords / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species Method / / / Resolution : 0.88 Å Authors Correy, G.J. / Young, I.D. / Thompson, M.C. / Fraser, J.S. Funding support Organization Grant number Country National Science Foundation (NSF, United States) 2031205
Journal : Sci Adv / Year : 2021Title : Fragment binding to the Nsp3 macrodomain of SARS-CoV-2 identified through crystallographic screening and computational docking.Authors: Schuller, M. / Correy, G.J. / Gahbauer, S. / Fearon, D. / Wu, T. / Diaz, R.E. / Young, I.D. / Carvalho Martins, L. / Smith, D.H. / Schulze-Gahmen, U. / Owens, T.W. / Deshpande, I. / Merz, G. ... Authors : Schuller, M. / Correy, G.J. / Gahbauer, S. / Fearon, D. / Wu, T. / Diaz, R.E. / Young, I.D. / Carvalho Martins, L. / Smith, D.H. / Schulze-Gahmen, U. / Owens, T.W. / Deshpande, I. / Merz, G.E. / Thwin, A.C. / Biel, J.T. / Peters, J.K. / Moritz, M. / Herrera, N. / Kratochvil, H.T. / Aimon, A. / Bennett, J.M. / Brandao Neto, J. / Cohen, A.E. / Dias, A. / Douangamath, A. / Dunnett, L. / Fedorov, O. / Ferla, M.P. / Fuchs, M.R. / Gorrie-Stone, T.J. / Holton, J.M. / Johnson, M.G. / Krojer, T. / Meigs, G. / Powell, A.J. / Rack, J.G.M. / Rangel, V.L. / Russi, S. / Skyner, R.E. / Smith, C.A. / Soares, A.S. / Wierman, J.L. / Zhu, K. / O'Brien, P. / Jura, N. / Ashworth, A. / Irwin, J.J. / Thompson, M.C. / Gestwicki, J.E. / von Delft, F. / Shoichet, B.K. / Fraser, J.S. / Ahel, I. History Deposition Nov 17, 2020 Deposition site / Processing site Revision 1.0 Dec 9, 2020 Provider / Type Revision 1.1 Jan 27, 2021 Group / Category / entity_name_com / Item / _entity_name_com.nameRevision 1.2 Jun 30, 2021 Group / Category / citation_authorItem _citation.country / _citation.journal_abbrev ... _citation.country / _citation.journal_abbrev / _citation.journal_id_CSD / _citation.journal_id_ISSN / _citation.journal_volume / _citation.pdbx_database_id_DOI / _citation.pdbx_database_id_PubMed / _citation.title / _citation.year Revision 1.3 Mar 6, 2024 Group / Database references / Category / chem_comp_bond / database_2Item / _database_2.pdbx_database_accession
Show all Show less
 Yorodumi
Yorodumi Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords VIRAL PROTEIN /
VIRAL PROTEIN /  Macrodomain /
Macrodomain /  ADP-ribose /
ADP-ribose /  SARS-CoV-2
SARS-CoV-2 Function and homology information
Function and homology information mRNA guanylyltransferase activity / 5'-3' RNA helicase activity /
mRNA guanylyltransferase activity / 5'-3' RNA helicase activity /  Lyases; Phosphorus-oxygen lyases / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of TBK1 activity / Assembly of the SARS-CoV-2 Replication-Transcription Complex (RTC) / Maturation of replicase proteins / ISG15-specific peptidase activity / Transcription of SARS-CoV-2 sgRNAs ...protein guanylyltransferase activity / RNA endonuclease activity, producing 3'-phosphomonoesters /
Lyases; Phosphorus-oxygen lyases / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of TBK1 activity / Assembly of the SARS-CoV-2 Replication-Transcription Complex (RTC) / Maturation of replicase proteins / ISG15-specific peptidase activity / Transcription of SARS-CoV-2 sgRNAs ...protein guanylyltransferase activity / RNA endonuclease activity, producing 3'-phosphomonoesters /  mRNA guanylyltransferase activity / 5'-3' RNA helicase activity /
mRNA guanylyltransferase activity / 5'-3' RNA helicase activity /  Lyases; Phosphorus-oxygen lyases / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of TBK1 activity / Assembly of the SARS-CoV-2 Replication-Transcription Complex (RTC) / Maturation of replicase proteins / ISG15-specific peptidase activity / Transcription of SARS-CoV-2 sgRNAs / Translation of Replicase and Assembly of the Replication Transcription Complex / TRAF3-dependent IRF activation pathway / Replication of the SARS-CoV-2 genome /
Lyases; Phosphorus-oxygen lyases / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of TBK1 activity / Assembly of the SARS-CoV-2 Replication-Transcription Complex (RTC) / Maturation of replicase proteins / ISG15-specific peptidase activity / Transcription of SARS-CoV-2 sgRNAs / Translation of Replicase and Assembly of the Replication Transcription Complex / TRAF3-dependent IRF activation pathway / Replication of the SARS-CoV-2 genome /  snRNP Assembly / double membrane vesicle viral factory outer membrane /
snRNP Assembly / double membrane vesicle viral factory outer membrane /  Hydrolases; Acting on ester bonds; Exoribonucleases producing 5'-phosphomonoesters /
Hydrolases; Acting on ester bonds; Exoribonucleases producing 5'-phosphomonoesters /  SARS coronavirus main proteinase / host cell endosome / 3'-5'-RNA exonuclease activity / host cell endoplasmic reticulum-Golgi intermediate compartment / : / symbiont-mediated suppression of host toll-like receptor signaling pathway / symbiont-mediated degradation of host mRNA /
SARS coronavirus main proteinase / host cell endosome / 3'-5'-RNA exonuclease activity / host cell endoplasmic reticulum-Golgi intermediate compartment / : / symbiont-mediated suppression of host toll-like receptor signaling pathway / symbiont-mediated degradation of host mRNA /  mRNA guanylyltransferase / symbiont-mediated suppression of host ISG15-protein conjugation / G-quadruplex RNA binding / SARS-CoV-2 modulates host translation machinery /
mRNA guanylyltransferase / symbiont-mediated suppression of host ISG15-protein conjugation / G-quadruplex RNA binding / SARS-CoV-2 modulates host translation machinery /  omega peptidase activity / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of IRF3 activity / mRNA (guanine-N7)-methyltransferase / host cell Golgi apparatus / methyltransferase cap1 / symbiont-mediated perturbation of host ubiquitin-like protein modification /
omega peptidase activity / symbiont-mediated suppression of host cytoplasmic pattern recognition receptor signaling pathway via inhibition of IRF3 activity / mRNA (guanine-N7)-methyltransferase / host cell Golgi apparatus / methyltransferase cap1 / symbiont-mediated perturbation of host ubiquitin-like protein modification /  mRNA (nucleoside-2'-O-)-methyltransferase activity /
mRNA (nucleoside-2'-O-)-methyltransferase activity /  DNA helicase / mRNA 5'-cap (guanine-N7-)-methyltransferase activity /
DNA helicase / mRNA 5'-cap (guanine-N7-)-methyltransferase activity /  ubiquitinyl hydrolase 1 / cysteine-type deubiquitinase activity /
ubiquitinyl hydrolase 1 / cysteine-type deubiquitinase activity /  Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases /
Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases /  single-stranded RNA binding / host cell endoplasmic reticulum membrane / host cell perinuclear region of cytoplasm /
single-stranded RNA binding / host cell endoplasmic reticulum membrane / host cell perinuclear region of cytoplasm /  viral protein processing /
viral protein processing /  lyase activity /
lyase activity /  RNA helicase / induction by virus of host autophagy / copper ion binding /
RNA helicase / induction by virus of host autophagy / copper ion binding /  RNA-directed RNA polymerase / symbiont-mediated suppression of host gene expression / viral RNA genome replication / cysteine-type endopeptidase activity /
RNA-directed RNA polymerase / symbiont-mediated suppression of host gene expression / viral RNA genome replication / cysteine-type endopeptidase activity /  RNA-dependent RNA polymerase activity / DNA-templated transcription /
RNA-dependent RNA polymerase activity / DNA-templated transcription /  lipid binding / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / host cell nucleus / SARS-CoV-2 activates/modulates innate and adaptive immune responses /
lipid binding / symbiont-mediated suppression of host type I interferon-mediated signaling pathway / host cell nucleus / SARS-CoV-2 activates/modulates innate and adaptive immune responses /  ATP hydrolysis activity /
ATP hydrolysis activity /  proteolysis /
proteolysis /  RNA binding / zinc ion binding /
RNA binding / zinc ion binding /  ATP binding /
ATP binding /  membrane
membrane
 Severe acute respiratory syndrome coronavirus 2
Severe acute respiratory syndrome coronavirus 2 X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 0.88 Å
MOLECULAR REPLACEMENT / Resolution: 0.88 Å  Authors
Authors United States, 1items
United States, 1items  Citation
Citation Journal: Sci Adv / Year: 2021
Journal: Sci Adv / Year: 2021 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 7kqp.cif.gz
7kqp.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb7kqp.ent.gz
pdb7kqp.ent.gz PDB format
PDB format 7kqp.json.gz
7kqp.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/kq/7kqp
https://data.pdbj.org/pub/pdb/validation_reports/kq/7kqp ftp://data.pdbj.org/pub/pdb/validation_reports/kq/7kqp
ftp://data.pdbj.org/pub/pdb/validation_reports/kq/7kqp Links
Links Assembly
Assembly


 Components
Components
 Severe acute respiratory syndrome coronavirus 2
Severe acute respiratory syndrome coronavirus 2
 Escherichia coli BL21(DE3) (bacteria)
Escherichia coli BL21(DE3) (bacteria) Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases
Hydrolases; Acting on peptide bonds (peptidases); Cysteine endopeptidases Water
Water X-RAY DIFFRACTION / Number of used crystals: 1
X-RAY DIFFRACTION / Number of used crystals: 1  Sample preparation
Sample preparation
 SYNCHROTRON / Site:
SYNCHROTRON / Site:  NSLS-II
NSLS-II  / Beamline: 17-ID-2 / Wavelength: 0.77492 Å
/ Beamline: 17-ID-2 / Wavelength: 0.77492 Å : 0.77492 Å / Relative weight: 1
: 0.77492 Å / Relative weight: 1  Processing
Processing :
:  MOLECULAR REPLACEMENT
MOLECULAR REPLACEMENT Movie
Movie Controller
Controller


























































































































































































































































 PDBj
PDBj