+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 6ygf | ||||||
|---|---|---|---|---|---|---|---|
| Title | NADase from Aspergillus fumigatus with trapped reaction products | ||||||
 Components Components | AfNADase | ||||||
 Keywords Keywords |  HYDROLASE / HYDROLASE /  NAD+ glycohydrolase / NAD / Ca-binding / NAD+ glycohydrolase / NAD / Ca-binding /  homodimer / homodimer /  glycoprotein / glycoprotein /  nicotinamide / nicotinamide /  adenosine diphosphate ribose adenosine diphosphate ribose | ||||||
| Function / homology |  Function and homology information Function and homology information: /  NAD+ glycohydrolase / NAD+ glycohydrolase /  Hydrolases; Glycosylases; Hydrolysing N-glycosyl compounds / extracellular region / Hydrolases; Glycosylases; Hydrolysing N-glycosyl compounds / extracellular region /  metal ion binding metal ion bindingSimilarity search - Function | ||||||
| Biological species |   Aspergillus fumigatus Af293 (mold) Aspergillus fumigatus Af293 (mold) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 1.7 Å MOLECULAR REPLACEMENT / Resolution: 1.7 Å | ||||||
 Authors Authors | Stromland, O. / Ziegler, M. / Kallio, J.P. | ||||||
 Citation Citation |  Journal: Nat Commun / Year: 2021 Journal: Nat Commun / Year: 2021Title: Discovery of fungal surface NADases predominantly present in pathogenic species. Authors: Stromland, O. / Kallio, J.P. / Pschibul, A. / Skoge, R.H. / Hardardottir, H.M. / Sverkeli, L.J. / Heinekamp, T. / Kniemeyer, O. / Migaud, M. / Makarov, M.V. / Gossmann, T.I. / Brakhage, A.A. / Ziegler, M. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  6ygf.cif.gz 6ygf.cif.gz | 240.9 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb6ygf.ent.gz pdb6ygf.ent.gz | 160 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  6ygf.json.gz 6ygf.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/yg/6ygf https://data.pdbj.org/pub/pdb/validation_reports/yg/6ygf ftp://data.pdbj.org/pub/pdb/validation_reports/yg/6ygf ftp://data.pdbj.org/pub/pdb/validation_reports/yg/6ygf | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 |
| ||||||||||||
| Unit cell |
|
- Components
Components
-Protein , 1 types, 2 molecules BA
| #1: Protein | Mass: 27680.092 Da / Num. of mol.: 2 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Aspergillus fumigatus Af293 (mold) / Gene: AFUA_6G14470 / Production host: Aspergillus fumigatus Af293 (mold) / Gene: AFUA_6G14470 / Production host:   Spodoptera frugiperda (fall armyworm) / References: UniProt: Q4WL81 Spodoptera frugiperda (fall armyworm) / References: UniProt: Q4WL81 |
|---|
-Sugars , 4 types, 6 molecules 
| #2: Polysaccharide |  / Mass: 367.349 Da / Num. of mol.: 2 / Mass: 367.349 Da / Num. of mol.: 2Source method: isolated from a genetically manipulated source #3: Polysaccharide | alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]beta-D-mannopyranose-(1-4)-2-acetamido-2- ...alpha-D-mannopyranose-(1-3)-[alpha-D-mannopyranose-(1-6)]beta-D-mannopyranose-(1-4)-2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta-D-glucopyranose |  / Mass: 1056.964 Da / Num. of mol.: 1 / Mass: 1056.964 Da / Num. of mol.: 1Source method: isolated from a genetically manipulated source #4: Polysaccharide | 2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta- ...2-acetamido-2-deoxy-beta-D-glucopyranose-(1-4)-[alpha-L-fucopyranose-(1-6)]2-acetamido-2-deoxy-beta-D-glucopyranose |  / Mass: 570.542 Da / Num. of mol.: 1 / Mass: 570.542 Da / Num. of mol.: 1Source method: isolated from a genetically manipulated source #5: Sugar |  N-Acetylglucosamine N-Acetylglucosamine |
|---|
-Non-polymers , 5 types, 567 molecules 
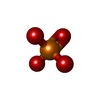
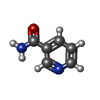
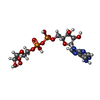

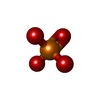



| #6: Chemical |  Glycerol Glycerol#7: Chemical |  Phosphate Phosphate#8: Chemical | ChemComp-NCA / |  Nicotinamide Nicotinamide#9: Chemical | ChemComp-AR6 / [( | #10: Water | ChemComp-HOH / |  Water Water |
|---|
-Details
| Has ligand of interest | Y |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.9 Å3/Da / Density % sol: 57.53 % |
|---|---|
Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop Details: 0.04 M Potassium phosphate monobasic, 16% PEG 8000 and 20% glycerol |
-Data collection
| Diffraction | Mean temperature: 100 K / Serial crystal experiment: N |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  PETRA III, DESY PETRA III, DESY  / Beamline: P11 / Wavelength: 1.0332 Å / Beamline: P11 / Wavelength: 1.0332 Å |
| Detector | Type: DECTRIS PILATUS 6M-F / Detector: PIXEL / Date: Oct 25, 2018 |
| Radiation | Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.0332 Å / Relative weight: 1 : 1.0332 Å / Relative weight: 1 |
| Reflection | Resolution: 1.7→85.9 Å / Num. obs: 68236 / % possible obs: 99.74 % / Redundancy: 2 % / Biso Wilson estimate: 27.48 Å2 / CC1/2: 0.999 / Rmerge(I) obs: 0.02465 / Net I/σ(I): 15.17 |
| Reflection shell | Resolution: 1.7→1.76 Å / Rmerge(I) obs: 0.644 / Mean I/σ(I) obs: 1.04 / Num. unique obs: 6579 / CC1/2: 0.477 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT MOLECULAR REPLACEMENTStarting model: D_1292105770 Resolution: 1.7→85.9 Å / SU ML: 0.2258 / Cross valid method: FREE R-VALUE / σ(F): 1.34 / Phase error: 19.8257
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Shrinkage radii: 0.9 Å / VDW probe radii: 1.11 Å | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 35.24 Å2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.7→85.9 Å
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LS refinement shell |
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS params. | Method: refined / Refine-ID: X-RAY DIFFRACTION
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement TLS group |
|
 Movie
Movie Controller
Controller















 PDBj
PDBj





