Entry Database : PDB / ID : 4akiTitle Dynein Motor Domain - LuAc derivative GLUTATHIONE S-TRANSFERASE CLASS-MU 26 KDA ISOZYME, DYNEIN HEAVY CHAIN CYTOPLASMIC Keywords / / / / / / / Function / homology Function Domain/homology Component
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / Biological species SCHISTOSOMA JAPONICUM (invertebrata)SACCHAROMYCES CEREVISIAE (brewer's yeast)Method / / / Resolution : 3.7 Å Authors Schmidt, H. / Gleave, E.S. / Carter, A.P. Journal : Nat.Struct.Mol.Biol. / Year : 2012Title : Insights Into Dynein Motor Domain Function from a 3.3 Angstrom Crystal StructureAuthors : Schmidt, H. / Gleave, E.S. / Carter, A.P. History Deposition Feb 22, 2012 Deposition site / Processing site Revision 1.0 Mar 14, 2012 Provider / Type Revision 1.1 May 16, 2012 Group Revision 1.2 Oct 21, 2015 Group Revision 1.3 Mar 15, 2017 Group Revision 1.4 May 8, 2024 Group Data collection / Database references ... Data collection / Database references / Derived calculations / Other Category chem_comp_atom / chem_comp_bond ... chem_comp_atom / chem_comp_bond / database_2 / pdbx_database_status / pdbx_struct_conn_angle / struct_conn / struct_site Item _database_2.pdbx_DOI / _database_2.pdbx_database_accession ... _database_2.pdbx_DOI / _database_2.pdbx_database_accession / _pdbx_database_status.status_code_sf / _pdbx_struct_conn_angle.ptnr1_auth_comp_id / _pdbx_struct_conn_angle.ptnr1_auth_seq_id / _pdbx_struct_conn_angle.ptnr1_label_asym_id / _pdbx_struct_conn_angle.ptnr1_label_atom_id / _pdbx_struct_conn_angle.ptnr1_label_comp_id / _pdbx_struct_conn_angle.ptnr1_label_seq_id / _pdbx_struct_conn_angle.ptnr3_auth_comp_id / _pdbx_struct_conn_angle.ptnr3_auth_seq_id / _pdbx_struct_conn_angle.ptnr3_label_asym_id / _pdbx_struct_conn_angle.ptnr3_label_atom_id / _pdbx_struct_conn_angle.ptnr3_label_comp_id / _pdbx_struct_conn_angle.ptnr3_label_seq_id / _pdbx_struct_conn_angle.value / _struct_conn.pdbx_dist_value / _struct_conn.ptnr1_auth_comp_id / _struct_conn.ptnr1_auth_seq_id / _struct_conn.ptnr1_label_asym_id / _struct_conn.ptnr1_label_atom_id / _struct_conn.ptnr1_label_comp_id / _struct_conn.ptnr1_label_seq_id / _struct_conn.ptnr2_auth_comp_id / _struct_conn.ptnr2_auth_seq_id / _struct_conn.ptnr2_label_asym_id / _struct_conn.ptnr2_label_atom_id / _struct_conn.ptnr2_label_comp_id / _struct_conn.ptnr2_label_seq_id / _struct_site.pdbx_auth_asym_id / _struct_site.pdbx_auth_comp_id / _struct_site.pdbx_auth_seq_id
Show all Show less Remark 650 HELIX DETERMINATION METHOD: AUTHOR PROVIDED. Remark 700 SHEET DETERMINATION METHOD: AUTHOR PROVIDED.
 Open data
Open data Basic information
Basic information Components
Components Keywords
Keywords MOTOR PROTEIN /
MOTOR PROTEIN /  DYNEIN /
DYNEIN /  DYNEIN HEAVY CHAIN /
DYNEIN HEAVY CHAIN /  DYNEIN MOTOR DOMAIN /
DYNEIN MOTOR DOMAIN /  MOTOR PROTEIN AAA+ PROTEIN / ASCE PROTEIN / P-LOOP NTPASE / CYTOSKELETAL MOTOR
MOTOR PROTEIN AAA+ PROTEIN / ASCE PROTEIN / P-LOOP NTPASE / CYTOSKELETAL MOTOR Function and homology information
Function and homology information karyogamy / establishment of mitotic spindle localization / astral microtubule / nuclear migration along microtubule / minus-end-directed microtubule motor activity /
karyogamy / establishment of mitotic spindle localization / astral microtubule / nuclear migration along microtubule / minus-end-directed microtubule motor activity /  cytoplasmic dynein complex / dynein light intermediate chain binding /
cytoplasmic dynein complex / dynein light intermediate chain binding /  spindle pole body / nuclear migration / dynein intermediate chain binding ...
spindle pole body / nuclear migration / dynein intermediate chain binding ... karyogamy / establishment of mitotic spindle localization / astral microtubule / nuclear migration along microtubule / minus-end-directed microtubule motor activity /
karyogamy / establishment of mitotic spindle localization / astral microtubule / nuclear migration along microtubule / minus-end-directed microtubule motor activity /  cytoplasmic dynein complex / dynein light intermediate chain binding /
cytoplasmic dynein complex / dynein light intermediate chain binding /  spindle pole body / nuclear migration / dynein intermediate chain binding / mitotic sister chromatid segregation / establishment of mitotic spindle orientation /
spindle pole body / nuclear migration / dynein intermediate chain binding / mitotic sister chromatid segregation / establishment of mitotic spindle orientation /  glutathione transferase /
glutathione transferase /  glutathione transferase activity / cytoplasmic microtubule / cytoplasmic microtubule organization / Neutrophil degranulation / mitotic spindle organization /
glutathione transferase activity / cytoplasmic microtubule / cytoplasmic microtubule organization / Neutrophil degranulation / mitotic spindle organization /  cell cortex /
cell cortex /  ATP hydrolysis activity /
ATP hydrolysis activity /  ATP binding /
ATP binding /  cytoplasm
cytoplasm
 SCHISTOSOMA JAPONICUM (invertebrata)
SCHISTOSOMA JAPONICUM (invertebrata)
 SACCHAROMYCES CEREVISIAE (brewer's yeast)
SACCHAROMYCES CEREVISIAE (brewer's yeast) X-RAY DIFFRACTION /
X-RAY DIFFRACTION /  SYNCHROTRON /
SYNCHROTRON /  SAD / Resolution: 3.7 Å
SAD / Resolution: 3.7 Å  Authors
Authors Citation
Citation Journal: Nat.Struct.Mol.Biol. / Year: 2012
Journal: Nat.Struct.Mol.Biol. / Year: 2012 Structure visualization
Structure visualization Molmil
Molmil Jmol/JSmol
Jmol/JSmol Downloads & links
Downloads & links Download
Download 4aki.cif.gz
4aki.cif.gz PDBx/mmCIF format
PDBx/mmCIF format pdb4aki.ent.gz
pdb4aki.ent.gz PDB format
PDB format 4aki.json.gz
4aki.json.gz PDBx/mmJSON format
PDBx/mmJSON format Other downloads
Other downloads https://data.pdbj.org/pub/pdb/validation_reports/ak/4aki
https://data.pdbj.org/pub/pdb/validation_reports/ak/4aki ftp://data.pdbj.org/pub/pdb/validation_reports/ak/4aki
ftp://data.pdbj.org/pub/pdb/validation_reports/ak/4aki Links
Links Assembly
Assembly
 Components
Components
 SCHISTOSOMA JAPONICUM (invertebrata), (gene. exp.)
SCHISTOSOMA JAPONICUM (invertebrata), (gene. exp.) 
 SACCHAROMYCES CEREVISIAE (brewer's yeast)
SACCHAROMYCES CEREVISIAE (brewer's yeast)
 SACCHAROMYCES CEREVISIAE (brewer's yeast)
SACCHAROMYCES CEREVISIAE (brewer's yeast) glutathione transferase
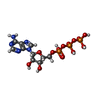
glutathione transferase Adenosine triphosphate
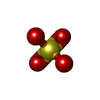
Adenosine triphosphate Sulfate
Sulfate X-RAY DIFFRACTION
X-RAY DIFFRACTION Sample preparation
Sample preparation SYNCHROTRON / Site:
SYNCHROTRON / Site:  Diamond
Diamond  / Beamline: I02 / Wavelength: 1.34072
/ Beamline: I02 / Wavelength: 1.34072  : 1.34072 Å / Relative weight: 1
: 1.34072 Å / Relative weight: 1  Processing
Processing :
:  SAD
SAD Movie
Movie Controller
Controller


























 PDBj
PDBj