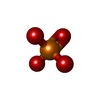+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 2egk | ||||||
|---|---|---|---|---|---|---|---|
| Title | Crystal Structure of Tamalin PDZ-Intrinsic Ligand Fusion Protein | ||||||
 Components Components | General receptor for phosphoinositides 1-associated scaffold protein | ||||||
 Keywords Keywords |  PROTEIN BINDING / PROTEIN BINDING /  PDZ domain / ligand fusion PDZ domain / ligand fusion | ||||||
| Function / homology |  Function and homology information Function and homology informationregulation of neurotransmitter receptor transport, endosome to postsynaptic membrane /  PDZ domain binding / Schaffer collateral - CA1 synapse / PDZ domain binding / Schaffer collateral - CA1 synapse /  small GTPase binding / small GTPase binding /  postsynaptic membrane / postsynaptic membrane /  postsynaptic density / glutamatergic synapse / perinuclear region of cytoplasm / postsynaptic density / glutamatergic synapse / perinuclear region of cytoplasm /  signal transduction / identical protein binding / signal transduction / identical protein binding /  plasma membrane plasma membraneSimilarity search - Function | ||||||
| Biological species |   Rattus norvegicus (Norway rat) Rattus norvegicus (Norway rat) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MAD / Resolution: 2.85 Å MAD / Resolution: 2.85 Å | ||||||
 Authors Authors | Sugi, T. / Oyama, T. / Muto, T. / Nakanishi, S. / Morikawa, K. / Jingami, H. | ||||||
 Citation Citation |  Journal: Embo J. / Year: 2007 Journal: Embo J. / Year: 2007Title: Crystal structures of autoinhibitory PDZ domain of Tamalin: implications for metabotropic glutamate receptor trafficking regulation Authors: Sugi, T. / Oyama, T. / Muto, T. / Nakanishi, S. / Morikawa, K. / Jingami, H. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  2egk.cif.gz 2egk.cif.gz | 77.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb2egk.ent.gz pdb2egk.ent.gz | 62.6 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  2egk.json.gz 2egk.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/eg/2egk https://data.pdbj.org/pub/pdb/validation_reports/eg/2egk ftp://data.pdbj.org/pub/pdb/validation_reports/eg/2egk ftp://data.pdbj.org/pub/pdb/validation_reports/eg/2egk | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| 3 |
| ||||||||
| Unit cell |
|
- Components
Components
| #1: Protein | Mass: 11277.422 Da / Num. of mol.: 4 / Fragment: PDZ domain, c-terminal peptode(Intrinsic ligand) / Mutation: C135A Source method: isolated from a genetically manipulated source Source: (gene. exp.)   Rattus norvegicus (Norway rat) / Tissue: brain Rattus norvegicus (Norway rat) / Tissue: brain / Plasmid: pGEX4T-1 / Species (production host): Escherichia coli / Production host: / Plasmid: pGEX4T-1 / Species (production host): Escherichia coli / Production host:   Escherichia coli BL21 (bacteria) / Strain (production host): BL21 / References: UniProt: Q8R4T5 Escherichia coli BL21 (bacteria) / Strain (production host): BL21 / References: UniProt: Q8R4T5#2: Chemical |  Phosphate Phosphate#3: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 3.86 Å3/Da / Density % sol: 68.13 % Description: THE STRUCTURE FACTOR FILE CONTAINS FRIEDEL PAIRS. |
|---|---|
Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 7.5 Details: 0.1M HEPES-Na, 0.8M sodium dihydrogen phosphate, 0.8M potassium dihydrogen phosphate, pH 7.5, VAPOR DIFFUSION, HANGING DROP, temperature 293K |
-Data collection
| Diffraction | Mean temperature: 298 K | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  SPring-8 SPring-8  / Beamline: BL38B1 / Wavelength: 0.97910, 0.97940, 0.99000 / Beamline: BL38B1 / Wavelength: 0.97910, 0.97940, 0.99000 | ||||||||||||
| Detector | Type: RIGAKU JUPITER 210 / Detector: CCD / Details: mirrors | ||||||||||||
| Radiation | Monochromator: Si 111 / Protocol: MAD / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||
| Radiation wavelength |
| ||||||||||||
| Reflection | Resolution: 2.85→50 Å / Num. all: 31406 / Num. obs: 29994 / % possible obs: 97.2 % / Observed criterion σ(F): 0 / Observed criterion σ(I): 0 / Redundancy: 3.7 % / Rmerge(I) obs: 0.108 / Net I/σ(I): 5.9 | ||||||||||||
| Reflection shell | Resolution: 2.85→2.95 Å / Redundancy: 2.8 % / Rmerge(I) obs: 0.396 / Num. unique all: 2729 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MAD / Resolution: 2.85→50 Å / Rfactor Rfree error: 0.009 / Data cutoff high absF: 199788.9 / Data cutoff low absF: 0 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: Engh & Huber / Details: THE FRIEDEL PAIRS WERE USED FOR PHASING. MAD / Resolution: 2.85→50 Å / Rfactor Rfree error: 0.009 / Data cutoff high absF: 199788.9 / Data cutoff low absF: 0 / Isotropic thermal model: RESTRAINED / Cross valid method: THROUGHOUT / σ(F): 0 / Stereochemistry target values: Engh & Huber / Details: THE FRIEDEL PAIRS WERE USED FOR PHASING.
| ||||||||||||||||||||||||||||||||||||
| Solvent computation | Solvent model: FLAT MODEL / Bsol: 41.879 Å2 / ksol: 0.350717 e/Å3 | ||||||||||||||||||||||||||||||||||||
| Displacement parameters |
| ||||||||||||||||||||||||||||||||||||
| Refine analyze |
| ||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.85→50 Å
| ||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
| ||||||||||||||||||||||||||||||||||||
| LS refinement shell | Resolution: 2.85→2.95 Å / Total num. of bins used: 6
| ||||||||||||||||||||||||||||||||||||
| Xplor file |
|
 Movie
Movie Controller
Controller















 PDBj
PDBj