+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1yc2 | ||||||
|---|---|---|---|---|---|---|---|
| Title | Sir2Af2-NAD-ADPribose-nicotinamide | ||||||
 Components Components | NAD-dependent deacetylase 2 | ||||||
 Keywords Keywords |  HYDROLASE / HYDROLASE /  sir2 / sir2 /  sirtuin / sirtuin /  nicotinamide / NAD / ADPribose / nicotinamide / NAD / ADPribose /  ternary complex ternary complex | ||||||
| Function / homology |  Function and homology information Function and homology informationprotein-malonyllysine demalonylase activity / protein-succinyllysine desuccinylase activity / NAD-dependent protein lysine deacetylase activity / protein acetyllysine N-acetyltransferase /  NAD+ binding / NAD+ binding /  transferase activity / zinc ion binding / transferase activity / zinc ion binding /  cytoplasm cytoplasmSimilarity search - Function | ||||||
| Biological species |    Archaeoglobus fulgidus (archaea) Archaeoglobus fulgidus (archaea) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MOLECULAR REPLACEMENT / Resolution: 2.4 Å MOLECULAR REPLACEMENT / Resolution: 2.4 Å | ||||||
 Authors Authors | Avalos, J.L. / Bever, M.K. / Wolberger, C. | ||||||
 Citation Citation |  Journal: Mol.Cell / Year: 2005 Journal: Mol.Cell / Year: 2005Title: Mechanism of Sirtuin Inhibition by Nicotinamide: Altering the NAD(+) Cosubstrate Specificity of a Sir2 Enzyme. Authors: Avalos, J.L. / Bever, K.M. / Wolberger, C. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1yc2.cif.gz 1yc2.cif.gz | 269.7 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1yc2.ent.gz pdb1yc2.ent.gz | 216.3 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1yc2.json.gz 1yc2.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/yc/1yc2 https://data.pdbj.org/pub/pdb/validation_reports/yc/1yc2 ftp://data.pdbj.org/pub/pdb/validation_reports/yc/1yc2 ftp://data.pdbj.org/pub/pdb/validation_reports/yc/1yc2 | HTTPS FTP |
|---|
-Related structure data
- Links
Links
- Assembly
Assembly
- Components
Components
-Protein , 1 types, 5 molecules ABCDE
| #1: Protein | Mass: 28537.006 Da / Num. of mol.: 5 Source method: isolated from a genetically manipulated source Source: (gene. exp.)    Archaeoglobus fulgidus (archaea) / Gene: npdA2 / Plasmid: pET11a / Production host: Archaeoglobus fulgidus (archaea) / Gene: npdA2 / Plasmid: pET11a / Production host:   Escherichia coli (E. coli) / Strain (production host): BL21(DE3)PLysS Escherichia coli (E. coli) / Strain (production host): BL21(DE3)PLysSReferences: UniProt: O30124,  Hydrolases; Acting on carbon-nitrogen bonds, other than peptide bonds; In linear amides Hydrolases; Acting on carbon-nitrogen bonds, other than peptide bonds; In linear amides |
|---|
-Non-polymers , 10 types, 287 molecules 
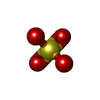
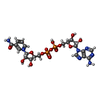
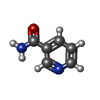
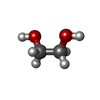

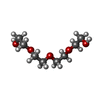
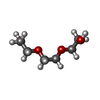
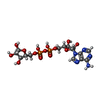










| #2: Chemical | ChemComp-ZN / #3: Chemical | ChemComp-SO4 /  Sulfate Sulfate#4: Chemical | ChemComp-NAD /  Nicotinamide adenine dinucleotide Nicotinamide adenine dinucleotide#5: Chemical | ChemComp-NCA /  Nicotinamide Nicotinamide#6: Chemical | ChemComp-EDO /  Ethylene glycol Ethylene glycol#7: Chemical |  Polyethylene glycol Polyethylene glycol#8: Chemical | ChemComp-PG4 / |  Polyethylene glycol Polyethylene glycol#9: Chemical | ChemComp-PGE / |  Polyethylene glycol Polyethylene glycol#10: Chemical | ChemComp-APR / | #11: Water | ChemComp-HOH / |  Water Water |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.6 Å3/Da / Density % sol: 52.7 % |
|---|---|
Crystal grow | Temperature: 293 K / Method: vapor diffusion, hanging drop / pH: 7.4 Details: HEPES, ammonium sulfate, PEG400, nicotinamide, pH 7.4, VAPOR DIFFUSION, HANGING DROP, temperature 293K |
-Data collection
| Diffraction | Mean temperature: 93 K |
|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  NSLS NSLS  / Beamline: X25 / Wavelength: 1.099997 Å / Beamline: X25 / Wavelength: 1.099997 Å |
| Detector | Type: ADSC QUANTUM 4 / Detector: CCD / Date: Apr 20, 2003 |
| Radiation | Monochromator: 2 CRYSTAL SI MONOCHROMATOR / Protocol: SINGLE WAVELENGTH / Monochromatic (M) / Laue (L): M / Scattering type: x-ray |
| Radiation wavelength | Wavelength : 1.099997 Å / Relative weight: 1 : 1.099997 Å / Relative weight: 1 |
| Reflection | Resolution: 2.4→30 Å / Num. obs: 59851 / % possible obs: 98.1 % / Observed criterion σ(F): 2 / Observed criterion σ(I): 2 / Redundancy: 7.1 % / Rmerge(I) obs: 0.108 / Rsym value: 0.086 / Net I/σ(I): 19.7 |
| Reflection shell | Resolution: 2.4→2.49 Å / % possible all: 82.1 |
- Processing
Processing
| Software |
| |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MOLECULAR REPLACEMENT / Resolution: 2.4→30 Å / σ(F): 0 / σ(I): 0 / Stereochemistry target values: Engh & Huber MOLECULAR REPLACEMENT / Resolution: 2.4→30 Å / σ(F): 0 / σ(I): 0 / Stereochemistry target values: Engh & Huber
| |||||||||||||||||||||||||
| Displacement parameters | Biso mean: 44.7 Å2 | |||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 2.4→30 Å
| |||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller














 PDBj
PDBj





















