+ Open data
Open data
- Basic information
Basic information
| Entry | Database: PDB / ID: 1uuj | ||||||
|---|---|---|---|---|---|---|---|
| Title | N-terminal domain of Lissencephaly-1 protein (Lis-1) | ||||||
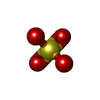
 Components Components | PLATELET-ACTIVATING FACTOR ACETYLHYDROLASE IB ALPHA SUBUNIT | ||||||
 Keywords Keywords |  CELL DIVISION / CELL DIVISION /  PLATELET-ACTIVATING FACTOR ACETYLHYDROLASE / PLATELET-ACTIVATING FACTOR ACETYLHYDROLASE /  MITOSIS / MITOSIS /  NEUROGENESIS / NEUROGENESIS /  CYTOSKELETON / CYTOSKELETON /  MICROTUBULE MICROTUBULE | ||||||
| Function / homology |  Function and homology information Function and homology informationpositive regulation of cellular component organization / : / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / Resolution of Sister Chromatid Cohesion / COPI-independent Golgi-to-ER retrograde traffic / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / RHO GTPases Activate Formins /  platelet-activating factor acetyltransferase activity / Loss of Nlp from mitotic centrosomes ...positive regulation of cellular component organization / : / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / Resolution of Sister Chromatid Cohesion / COPI-independent Golgi-to-ER retrograde traffic / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / RHO GTPases Activate Formins / platelet-activating factor acetyltransferase activity / Loss of Nlp from mitotic centrosomes ...positive regulation of cellular component organization / : / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / Resolution of Sister Chromatid Cohesion / COPI-independent Golgi-to-ER retrograde traffic / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / RHO GTPases Activate Formins /  platelet-activating factor acetyltransferase activity / Loss of Nlp from mitotic centrosomes / Recruitment of mitotic centrosome proteins and complexes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / AURKA Activation by TPX2 / corpus callosum morphogenesis / establishment of planar polarity of embryonic epithelium / microtubule cytoskeleton organization involved in establishment of planar polarity / platelet-activating factor acetyltransferase activity / Loss of Nlp from mitotic centrosomes / Recruitment of mitotic centrosome proteins and complexes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / AURKA Activation by TPX2 / corpus callosum morphogenesis / establishment of planar polarity of embryonic epithelium / microtubule cytoskeleton organization involved in establishment of planar polarity /  Regulation of PLK1 Activity at G2/M Transition / ameboidal-type cell migration / Separation of Sister Chromatids / interneuron migration / Regulation of PLK1 Activity at G2/M Transition / ameboidal-type cell migration / Separation of Sister Chromatids / interneuron migration /  1-alkyl-2-acetylglycerophosphocholine esterase complex / maintenance of centrosome location / microtubule sliding / COPI-independent Golgi-to-ER retrograde traffic / 1-alkyl-2-acetylglycerophosphocholine esterase complex / maintenance of centrosome location / microtubule sliding / COPI-independent Golgi-to-ER retrograde traffic /  acrosome assembly / radial glia-guided pyramidal neuron migration / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / microtubule organizing center organization / cerebral cortex neuron differentiation / central region of growth cone / positive regulation of embryonic development / reelin-mediated signaling pathway / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / Resolution of Sister Chromatid Cohesion / establishment of centrosome localization / positive regulation of cytokine-mediated signaling pathway / cortical microtubule organization / astral microtubule / RHO GTPases Activate Formins / layer formation in cerebral cortex / nuclear membrane disassembly / Separation of Sister Chromatids / auditory receptor cell development / Loss of Nlp from mitotic centrosomes / Recruitment of mitotic centrosome proteins and complexes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / positive regulation of dendritic spine morphogenesis / AURKA Activation by TPX2 / vesicle transport along microtubule / acrosome assembly / radial glia-guided pyramidal neuron migration / Amplification of signal from unattached kinetochores via a MAD2 inhibitory signal / microtubule organizing center organization / cerebral cortex neuron differentiation / central region of growth cone / positive regulation of embryonic development / reelin-mediated signaling pathway / Mitotic Prometaphase / EML4 and NUDC in mitotic spindle formation / Resolution of Sister Chromatid Cohesion / establishment of centrosome localization / positive regulation of cytokine-mediated signaling pathway / cortical microtubule organization / astral microtubule / RHO GTPases Activate Formins / layer formation in cerebral cortex / nuclear membrane disassembly / Separation of Sister Chromatids / auditory receptor cell development / Loss of Nlp from mitotic centrosomes / Recruitment of mitotic centrosome proteins and complexes / Loss of proteins required for interphase microtubule organization from the centrosome / Recruitment of NuMA to mitotic centrosomes / Anchoring of the basal body to the plasma membrane / positive regulation of dendritic spine morphogenesis / AURKA Activation by TPX2 / vesicle transport along microtubule /  Regulation of PLK1 Activity at G2/M Transition / stem cell division / stereocilium / myeloid leukocyte migration / microtubule plus-end binding / negative regulation of JNK cascade / retrograde axonal transport / brain morphogenesis / Regulation of PLK1 Activity at G2/M Transition / stem cell division / stereocilium / myeloid leukocyte migration / microtubule plus-end binding / negative regulation of JNK cascade / retrograde axonal transport / brain morphogenesis /  motile cilium / nuclear migration / osteoclast development / microtubule associated complex / regulation of postsynapse organization / motile cilium / nuclear migration / osteoclast development / microtubule associated complex / regulation of postsynapse organization /  kinesin complex / dynein intermediate chain binding / dynein complex binding / cochlea development / kinesin complex / dynein intermediate chain binding / dynein complex binding / cochlea development /  regulation of GTPase activity / transmission of nerve impulse / cytoplasmic microtubule / cell leading edge / germ cell development / establishment of mitotic spindle orientation / neuromuscular process controlling balance / regulation of GTPase activity / transmission of nerve impulse / cytoplasmic microtubule / cell leading edge / germ cell development / establishment of mitotic spindle orientation / neuromuscular process controlling balance /  protein secretion / neuroblast proliferation / positive regulation of axon extension / microtubule-based process / lipid catabolic process / regulation of microtubule cytoskeleton organization / axon cytoplasm / JNK cascade / positive regulation of mitotic cell cycle / adult locomotory behavior / establishment of localization in cell / hippocampus development / protein secretion / neuroblast proliferation / positive regulation of axon extension / microtubule-based process / lipid catabolic process / regulation of microtubule cytoskeleton organization / axon cytoplasm / JNK cascade / positive regulation of mitotic cell cycle / adult locomotory behavior / establishment of localization in cell / hippocampus development /  phosphoprotein binding / phosphoprotein binding /  brain development / brain development /  neuron migration / Schaffer collateral - CA1 synapse / modulation of chemical synaptic transmission / neuron migration / Schaffer collateral - CA1 synapse / modulation of chemical synaptic transmission /  kinetochore / cerebral cortex development / microtubule cytoskeleton organization / microtubule cytoskeleton / kinetochore / cerebral cortex development / microtubule cytoskeleton organization / microtubule cytoskeleton /  cell migration cell migrationSimilarity search - Function | ||||||
| Biological species |   MUS MUSCULUS (house mouse) MUS MUSCULUS (house mouse) | ||||||
| Method |  X-RAY DIFFRACTION / X-RAY DIFFRACTION /  SYNCHROTRON / SYNCHROTRON /  MAD / Resolution: 1.75 Å MAD / Resolution: 1.75 Å | ||||||
 Authors Authors | Cooper, D.R. / Kim, M.H. / Devedjiev, Y. / Derewenda, U. / Derewenda, Z.S. | ||||||
 Citation Citation |  Journal: Structure / Year: 2004 Journal: Structure / Year: 2004Title: The Structure of the N-Terminal Domain of the Product of the Lissencephaly Gene Lis1 and its Functional Implications Authors: Kim, M.H. / Cooper, D.R. / Oleksy, A. / Devedjiev, Y. / Derewenda, U. / Reiner, O. / Otlewski, J. / Derewenda, Z.S. | ||||||
| History |
|
- Structure visualization
Structure visualization
| Structure viewer | Molecule:  Molmil Molmil Jmol/JSmol Jmol/JSmol |
|---|
- Downloads & links
Downloads & links
- Download
Download
| PDBx/mmCIF format |  1uuj.cif.gz 1uuj.cif.gz | 76.6 KB | Display |  PDBx/mmCIF format PDBx/mmCIF format |
|---|---|---|---|---|
| PDB format |  pdb1uuj.ent.gz pdb1uuj.ent.gz | 63.7 KB | Display |  PDB format PDB format |
| PDBx/mmJSON format |  1uuj.json.gz 1uuj.json.gz | Tree view |  PDBx/mmJSON format PDBx/mmJSON format | |
| Others |  Other downloads Other downloads |
-Validation report
| Arichive directory |  https://data.pdbj.org/pub/pdb/validation_reports/uu/1uuj https://data.pdbj.org/pub/pdb/validation_reports/uu/1uuj ftp://data.pdbj.org/pub/pdb/validation_reports/uu/1uuj ftp://data.pdbj.org/pub/pdb/validation_reports/uu/1uuj | HTTPS FTP |
|---|
-Related structure data
| Similar structure data |
|---|
- Links
Links
- Assembly
Assembly
| Deposited unit | 
| ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 1 | 
| ||||||||
| 2 | 
| ||||||||
| Unit cell |
|
- Components
Components
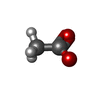
| #1: Protein | Mass: 10272.154 Da / Num. of mol.: 4 / Fragment: N-TERMINAL DOMAIN RESIDUES 1-85 Source method: isolated from a genetically manipulated source Source: (gene. exp.)   MUS MUSCULUS (house mouse) / Organ: BRAIN MUS MUSCULUS (house mouse) / Organ: BRAIN / Production host: / Production host:   ESCHERICHIA COLI (E. coli) / Strain (production host): B834 (DE3) / References: UniProt: P43035, UniProt: P63005*PLUS ESCHERICHIA COLI (E. coli) / Strain (production host): B834 (DE3) / References: UniProt: P43035, UniProt: P63005*PLUS#2: Chemical | ChemComp-SO4 /  Sulfate Sulfate#3: Chemical | ChemComp-ACT / |  Acetate Acetate#4: Chemical | ChemComp-BEZ / |  Benzoic acid Benzoic acid#5: Water | ChemComp-HOH / |  Water WaterSequence details | INITIAL 2 RESIDUES ARE CLONING ARTIFACTS. | |
|---|
-Experimental details
-Experiment
| Experiment | Method:  X-RAY DIFFRACTION / Number of used crystals: 1 X-RAY DIFFRACTION / Number of used crystals: 1 |
|---|
- Sample preparation
Sample preparation
| Crystal | Density Matthews: 2.1 Å3/Da / Density % sol: 49.6 % |
|---|---|
Crystal grow | Method: microbatch / pH: 4.5 Details: CRYSTALS WERE GROWN USING SITTING-DROP VAPOUR-DIFFUSION UNDER MINERAL OIL USING A 1:1 MIXTURE OF PROTEIN AND 1.7 M (NH4)2SO4 AND 0.1 M NA3-CITRATE, PH 4.5 |
-Data collection
| Diffraction | Mean temperature: 100 K | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Diffraction source | Source:  SYNCHROTRON / Site: SYNCHROTRON / Site:  APS APS  / Beamline: 22-ID / Wavelength: 0.979392,0.979528, 0.964216 / Beamline: 22-ID / Wavelength: 0.979392,0.979528, 0.964216 | ||||||||||||
| Detector | Type: MARRESEARCH / Detector: CCD / Date: Aug 22, 2003 | ||||||||||||
| Radiation | Protocol: MAD / Monochromatic (M) / Laue (L): M / Scattering type: x-ray | ||||||||||||
| Radiation wavelength |
| ||||||||||||
| Reflection | Resolution: 1.75→50 Å / Num. obs: 33378 / % possible obs: 96.1 % / Redundancy: 12 % / Biso Wilson estimate: 26.88 Å2 / Rmerge(I) obs: 0.057 / Net I/σ(I): 40 | ||||||||||||
| Reflection shell | Resolution: 1.75→1.81 Å / Redundancy: 9 % / Rmerge(I) obs: 0.477 / Mean I/σ(I) obs: 3 / % possible all: 78.9 |
- Processing
Processing
| Software |
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Refinement | Method to determine structure : :  MAD / Resolution: 1.75→40 Å / Cor.coef. Fo:Fc: 0.955 / Cor.coef. Fo:Fc free: 0.924 / SU B: 2.377 / SU ML: 0.077 / TLS residual ADP flag: LIKELY RESIDUAL / Cross valid method: THROUGHOUT / ESU R: 0.125 / ESU R Free: 0.131 MAD / Resolution: 1.75→40 Å / Cor.coef. Fo:Fc: 0.955 / Cor.coef. Fo:Fc free: 0.924 / SU B: 2.377 / SU ML: 0.077 / TLS residual ADP flag: LIKELY RESIDUAL / Cross valid method: THROUGHOUT / ESU R: 0.125 / ESU R Free: 0.131 Stereochemistry target values: MAXIMUM LIKELIHOOD WITH PHASES Details: HYDROGENS HAVE BEEN ADDED IN THE RIDING POSITIONS
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Solvent computation | Ion probe radii: 0.8 Å / Shrinkage radii: 0.8 Å / VDW probe radii: 1.4 Å / Solvent model: BABINET MODEL WITH MASK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Displacement parameters | Biso mean: 20.83 Å2
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refinement step | Cycle: LAST / Resolution: 1.75→40 Å
| ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Refine LS restraints |
|
 Movie
Movie Controller
Controller













 PDBj
PDBj