+ データを開く
データを開く
- 基本情報
基本情報
| 登録情報 |  | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| タイトル | Kir6.2-Q52R/SUR1 apo closed channel | |||||||||
 マップデータ マップデータ | Map of Kir6.2-Q52R SUR1 in an apo closed CTD-down, reconstructed with 12,320 particle C1 local reconstruction. | |||||||||
 試料 試料 |
| |||||||||
 キーワード キーワード |  ATP-sensitive potassium channel (ATP感受性カリウムチャネル) / ATP-sensitive potassium channel (ATP感受性カリウムチャネル) /  KATP channel (ATP感受性カリウムチャネル) / KATP channel (ATP感受性カリウムチャネル) /  SUR1 / Kir6.2-Q52R / potassium transport / metabolic sensor / SUR1 / Kir6.2-Q52R / potassium transport / metabolic sensor /  diabetes (糖尿病) / diabetes (糖尿病) /  phospholipid binding (リン脂質) / phospholipid binding (リン脂質) /  PIP2 (ホスファチジルイノシトール4,5-ビスリン酸) / PIP2 (ホスファチジルイノシトール4,5-ビスリン酸) /  TRANSPORT PROTEIN (運搬体タンパク質) TRANSPORT PROTEIN (運搬体タンパク質) | |||||||||
| 機能・相同性 |  機能・相同性情報 機能・相同性情報Regulation of insulin secretion /  ATP感受性カリウムチャネル / ABC-family proteins mediated transport / response to resveratrol / ATP-activated inward rectifier potassium channel activity / ventricular cardiac muscle tissue development / inward rectifying potassium channel / cell body fiber / ATP感受性カリウムチャネル / ABC-family proteins mediated transport / response to resveratrol / ATP-activated inward rectifier potassium channel activity / ventricular cardiac muscle tissue development / inward rectifying potassium channel / cell body fiber /  sulfonylurea receptor activity / CAMKK-AMPK signaling cascade ...Regulation of insulin secretion / sulfonylurea receptor activity / CAMKK-AMPK signaling cascade ...Regulation of insulin secretion /  ATP感受性カリウムチャネル / ABC-family proteins mediated transport / response to resveratrol / ATP-activated inward rectifier potassium channel activity / ventricular cardiac muscle tissue development / inward rectifying potassium channel / cell body fiber / ATP感受性カリウムチャネル / ABC-family proteins mediated transport / response to resveratrol / ATP-activated inward rectifier potassium channel activity / ventricular cardiac muscle tissue development / inward rectifying potassium channel / cell body fiber /  sulfonylurea receptor activity / CAMKK-AMPK signaling cascade / voltage-gated monoatomic ion channel activity involved in regulation of presynaptic membrane potential / sulfonylurea receptor activity / CAMKK-AMPK signaling cascade / voltage-gated monoatomic ion channel activity involved in regulation of presynaptic membrane potential /  inward rectifier potassium channel activity / ATPase-coupled monoatomic cation transmembrane transporter activity / inward rectifier potassium channel activity / ATPase-coupled monoatomic cation transmembrane transporter activity /  nervous system process / regulation of monoatomic ion transmembrane transport / inorganic cation transmembrane transport / nervous system process / regulation of monoatomic ion transmembrane transport / inorganic cation transmembrane transport /  ankyrin binding / ankyrin binding /  活動電位 / Ion homeostasis / response to ATP / response to testosterone / potassium ion import across plasma membrane / 活動電位 / Ion homeostasis / response to ATP / response to testosterone / potassium ion import across plasma membrane /  voltage-gated potassium channel activity / regulation of insulin secretion / voltage-gated potassium channel activity / regulation of insulin secretion /  介在板 / 介在板 /  axolemma / negative regulation of insulin secretion / ABC-type transporter activity / potassium ion transmembrane transport / axolemma / negative regulation of insulin secretion / ABC-type transporter activity / potassium ion transmembrane transport /  横行小管 / 横行小管 /  heat shock protein binding / acrosomal vesicle / heat shock protein binding / acrosomal vesicle /  regulation of membrane potential / response to ischemia / determination of adult lifespan / cellular response to glucose stimulus / positive regulation of protein localization to plasma membrane / potassium ion transport / regulation of membrane potential / response to ischemia / determination of adult lifespan / cellular response to glucose stimulus / positive regulation of protein localization to plasma membrane / potassium ion transport /  筋鞘 / cellular response to nicotine / glucose metabolic process / response to estradiol / 筋鞘 / cellular response to nicotine / glucose metabolic process / response to estradiol /  核膜 / 核膜 /  presynaptic membrane / cellular response to tumor necrosis factor / transmembrane transporter binding / response to hypoxia / presynaptic membrane / cellular response to tumor necrosis factor / transmembrane transporter binding / response to hypoxia /  エンドソーム / response to xenobiotic stimulus / neuronal cell body / glutamatergic synapse / apoptotic process / エンドソーム / response to xenobiotic stimulus / neuronal cell body / glutamatergic synapse / apoptotic process /  ATP hydrolysis activity / ATP hydrolysis activity /  ATP binding / ATP binding /  細胞膜 細胞膜類似検索 - 分子機能 | |||||||||
| 生物種 |   Rattus norvegicus (ドブネズミ) / Rattus norvegicus (ドブネズミ) /   Mesocricetus auratus (ゴールデンハムスター) Mesocricetus auratus (ゴールデンハムスター) | |||||||||
| 手法 |  単粒子再構成法 / 単粒子再構成法 /  クライオ電子顕微鏡法 / 解像度: 6.9 Å クライオ電子顕微鏡法 / 解像度: 6.9 Å | |||||||||
 データ登録者 データ登録者 | Driggers CM / Shyng S-L | |||||||||
| 資金援助 |  米国, 2件 米国, 2件
| |||||||||
 引用 引用 |  ジャーナル: Nat Commun / 年: 2024 ジャーナル: Nat Commun / 年: 2024タイトル: Structure of an open K channel reveals tandem PIP binding sites mediating the Kir6.2 and SUR1 regulatory interface. 著者: Camden M Driggers / Yi-Ying Kuo / Phillip Zhu / Assmaa ElSheikh / Show-Ling Shyng /   要旨: ATP-sensitive potassium (K) channels, composed of four pore-lining Kir6.2 subunits and four regulatory sulfonylurea receptor 1 (SUR1) subunits, control insulin secretion in pancreatic β-cells. K ...ATP-sensitive potassium (K) channels, composed of four pore-lining Kir6.2 subunits and four regulatory sulfonylurea receptor 1 (SUR1) subunits, control insulin secretion in pancreatic β-cells. K channel opening is stimulated by PIP and inhibited by ATP. Mutations that increase channel opening by PIP reduce ATP inhibition and cause neonatal diabetes. Although considerable evidence has implicated a role for PIP in K channel function, previously solved open-channel structures have lacked bound PIP, and mechanisms by which PIP regulates K channels remain unresolved. Here, we report the cryoEM structure of a K channel harboring the neonatal diabetes mutation Kir6.2-Q52R, in the open conformation, bound to amphipathic molecules consistent with natural C18:0/C20:4 long-chain PI(4,5)P at two adjacent binding sites between SUR1 and Kir6.2. The canonical PIP binding site is conserved among PIP-gated Kir channels. The non-canonical PIP binding site forms at the interface of Kir6.2 and SUR1. Functional studies demonstrate both binding sites determine channel activity. Kir6.2 pore opening is associated with a twist of the Kir6.2 cytoplasmic domain and a rotation of the N-terminal transmembrane domain of SUR1, which widens the inhibitory ATP binding pocket to disfavor ATP binding. The open conformation is particularly stabilized by the Kir6.2-Q52R residue through cation-π bonding with SUR1-W51. Together, these results uncover the cooperation between SUR1 and Kir6.2 in PIP binding and gating, explain the antagonistic regulation of K channels by PIP and ATP, and provide a putative mechanism by which Kir6.2-Q52R stabilizes an open channel to cause neonatal diabetes. | |||||||||
| 履歴 |
|
- 構造の表示
構造の表示
| 添付画像 |
|---|
- ダウンロードとリンク
ダウンロードとリンク
-EMDBアーカイブ
| マップデータ |  emd_43766.map.gz emd_43766.map.gz | 402.2 MB |  EMDBマップデータ形式 EMDBマップデータ形式 | |
|---|---|---|---|---|
| ヘッダ (付随情報) |  emd-43766-v30.xml emd-43766-v30.xml emd-43766.xml emd-43766.xml | 22.8 KB 22.8 KB | 表示 表示 |  EMDBヘッダ EMDBヘッダ |
| 画像 |  emd_43766.png emd_43766.png | 130.3 KB | ||
| Filedesc metadata |  emd-43766.cif.gz emd-43766.cif.gz | 7.4 KB | ||
| その他 |  emd_43766_half_map_1.map.gz emd_43766_half_map_1.map.gz emd_43766_half_map_2.map.gz emd_43766_half_map_2.map.gz | 764.4 MB 764.3 MB | ||
| アーカイブディレクトリ |  http://ftp.pdbj.org/pub/emdb/structures/EMD-43766 http://ftp.pdbj.org/pub/emdb/structures/EMD-43766 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43766 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43766 | HTTPS FTP |
-関連構造データ
- リンク
リンク
| EMDBのページ |  EMDB (EBI/PDBe) / EMDB (EBI/PDBe) /  EMDataResource EMDataResource |
|---|---|
| 「今月の分子」の関連する項目 |
- マップ
マップ
| ファイル |  ダウンロード / ファイル: emd_43766.map.gz / 形式: CCP4 / 大きさ: 824 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) ダウンロード / ファイル: emd_43766.map.gz / 形式: CCP4 / 大きさ: 824 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Map of Kir6.2-Q52R SUR1 in an apo closed CTD-down, reconstructed with 12,320 particle C1 local reconstruction. | ||||||||||||||||||||
| ボクセルのサイズ | X=Y=Z: 0.826 Å | ||||||||||||||||||||
| 密度 |
| ||||||||||||||||||||
| 対称性 | 空間群: 1 | ||||||||||||||||||||
| 詳細 | EMDB XML:
|
-添付データ
-ハーフマップ: Half map A of Kir6.2-Q52R SUR1 in an...
| ファイル | emd_43766_half_map_1.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map A of Kir6.2-Q52R SUR1 in an apo closed CTD-down, reconstructed with 12,320 particle C1 local reconstruction. | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
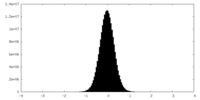
| 密度ヒストグラム |
-ハーフマップ: Half map B of Kir6.2-Q52R SUR1 in an...
| ファイル | emd_43766_half_map_2.map | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 注釈 | Half map B of Kir6.2-Q52R SUR1 in an apo closed CTD-down, reconstructed with 12,320 particle C1 local reconstruction. | ||||||||||||
| 投影像・断面図 |
| ||||||||||||
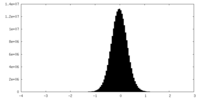
| 密度ヒストグラム |
- 試料の構成要素
試料の構成要素
-全体 : Kir6.2-Q52R/SUR1 open channel
| 全体 | 名称: Kir6.2-Q52R/SUR1 open channel |
|---|---|
| 要素 |
|
-超分子 #1: Kir6.2-Q52R/SUR1 open channel
| 超分子 | 名称: Kir6.2-Q52R/SUR1 open channel / タイプ: complex / ID: 1 / 親要素: 0 / 含まれる分子: all 詳細: Kir6.2-Q52R/SUR1 open KATP channel in the open conformation in complex with PIP2 and other phospholipids |
|---|---|
| 由来(天然) | 生物種:   Rattus norvegicus (ドブネズミ) Rattus norvegicus (ドブネズミ) |
| 分子量 | 理論値: 880 KDa |
-分子 #1: Kir6.2, Potassium inwardly rectifying channel, subfamily J, member 11
| 分子 | 名称: Kir6.2, Potassium inwardly rectifying channel, subfamily J, member 11 タイプ: protein_or_peptide / ID: 1 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:   Rattus norvegicus (ドブネズミ) / 器官: Pancreas / 細胞: Beta cell Rattus norvegicus (ドブネズミ) / 器官: Pancreas / 細胞: Beta cell |
| 組換発現 | 生物種:   Chlorocebus aethiops (ミドリザル) Chlorocebus aethiops (ミドリザル) |
| 配列 | 文字列: MLSRKGIIPE EYVLTRLAED PTEPRYRTRE RRARFVSKKG NCNVAHKNIR ERGRFLQDVF TTLVDLKWPH TLLIFTMSFL CSWLLFAMV WWLIAFAHGD LAPGEGTNVP CVTSIHSFSS AFLFSIEVQV TIGFGGRMVT EECPLAILIL IVQNIVGLMI N AIMLGCIF ...文字列: MLSRKGIIPE EYVLTRLAED PTEPRYRTRE RRARFVSKKG NCNVAHKNIR ERGRFLQDVF TTLVDLKWPH TLLIFTMSFL CSWLLFAMV WWLIAFAHGD LAPGEGTNVP CVTSIHSFSS AFLFSIEVQV TIGFGGRMVT EECPLAILIL IVQNIVGLMI N AIMLGCIF MKTAQAHRRA ETLIFSKHAV ITLRHGRLCF MLRVGDLRKS MIISATIHMQ VVRKTTSPEG EVVPLHQVDI PM ENGVGGN SIFLVAPLII YHVIDSNSPL YDLAPSDLHH HQDLEIIVIL EGVVETTGIT TQARTSYLAD EILWGQRFVP IVA EEDGRY SVDYSKFGNT VKVPTPLCTA RQLDEDRSLL DALTLASSRG PLRKRSVAVA KAKPKFSISP DSLS UniProtKB: ATP-sensitive inward rectifier potassium channel 11 |
-分子 #2: SUR1, ATP-binding cassette sub-family C member 8
| 分子 | 名称: SUR1, ATP-binding cassette sub-family C member 8 / タイプ: protein_or_peptide / ID: 2 / 光学異性体: LEVO |
|---|---|
| 由来(天然) | 生物種:   Mesocricetus auratus (ゴールデンハムスター) Mesocricetus auratus (ゴールデンハムスター) |
| 組換発現 | 生物種:   Chlorocebus aethiops (ミドリザル) Chlorocebus aethiops (ミドリザル) |
| 配列 | 文字列: MPLAFCGTEN HSAAYRVDQG VLNNGCFVDA LNVVPHVFLL FITFPILFIG WGSQSSKVHI HHSTWLHFPG HNLRWILTFI LLFVLVCEI AEGILSDGVT ESRHLHLYMP AGMAFMAAIT SVVYYHNIET SNFPKLLIAL LIYWTLAFIT KTIKFVKFYD H AIGFSQLR ...文字列: MPLAFCGTEN HSAAYRVDQG VLNNGCFVDA LNVVPHVFLL FITFPILFIG WGSQSSKVHI HHSTWLHFPG HNLRWILTFI LLFVLVCEI AEGILSDGVT ESRHLHLYMP AGMAFMAAIT SVVYYHNIET SNFPKLLIAL LIYWTLAFIT KTIKFVKFYD H AIGFSQLR FCLTGLLVIL YGMLLLVEVN VIRVRRYIFF KTPREVKPPE DLQDLGVRFL QPFVNLLSKG TYWWMNAFIK TA HKKPIDL RAIGKLPIAM RALTNYQRLC VAFDAQARKD TQSPQGARAI WRALCHAFGR RLILSSTFRI LADLLGFAGP LCI FGIVDH LGKENHVFQP KTQFLGVYFV SSQEFLGNAY VLAVLLFLAL LLQRTFLQAS YYVAIETGIN LRGAIQTKIY NKIM HLSTS NLSMGEMTAG QICNLVAIDT NQLMWFFFLC PNLWAMPVQI IVGVILLYYI LGVSALIGAA VIILLAPVQY FVATK LSQA QRSTLEHSNE RLKQTNEMLR GMKLLKLYAW ESIFCSRVEV TRRKEMTSLR AFAVYTSISI FMNTAIPIAA VLITFV GHV SFFKESDLSP SVAFASLSLF HILVTPLFLL SSVVRSTVKA LVSVQKLSEF LSSAEIREEQ CAPREPAPQG QAGKYQA VP LKVVNRKRPA REEVRDLLGP LQRLAPSMDG DADNFCVQII GGFFTWTPDG IPTLSNITIR IPRGQLTMIV GQVGCGKS S LLLATLGEMQ KVSGAVFWNS NLPDSEGEDP SSPERETAAG SDIRSRGPVA YASQKPWLLN ATVEENITFE SPFNKQRYK MVIEACSLQP DIDILPHGDQ TQIGERGINL SGGQRQRISV ARALYQQTNV VFLDDPFSAL DVHLSDHLMQ AGILELLRDD KRTVVLVTH KLQYLPHADW IIAMKDGTIQ REGTLKDFQR SECQLFEHWK TLMNRQDQEL EKETVMERKA SEPSQGLPRA M SSRDGLLL DEEEEEEEAA ESEEDDNLSS VLHQRAKIPW RACTKYLSSA GILLLSLLVF SQLLKHMVLV AIDYWLAKWT DS ALVLSPA ARNCSLSQEC DLDQSVYAMV FTLLCSLGIV LCLVTSVTVE WTGLKVAKRL HRSLLNRIIL APMRFFETTP LGS ILNRFS SDCNTIDQHI PSTLECLSRS TLLCVSALTV ISYVTPVFLV ALLPLAVVCY FIQKYFRVAS RDLQQLDDTT QLPL LSHFA ETVEGLTTIR AFRYEARFQQ KLLEYTDSNN IASLFLTAAN RWLEVRMEYI GACVVLIAAA TSISNSLHRE LSAGL VGLG LTYALMVSNY LNWMVRNLAD MEIQLGAVKR IHALLKTEAE SYEGLLAPSL IPKNWPDQGK IQIQNLSVRY DSSLKP VLK HVNALISPGQ KIGICGRTGS GKSSFSLAFF RMVDMFEGRI IIDGIDIAKL PLHTLRSRLS IILQDPVLFS GTIRFNL DP EKKCSDSTLW EALEIAQLKL VVKALPGGLD AIITEGGENF SQGQRQLFCL ARAFVRKTSI FIMDEATASI DMATENIL Q KVVMTAFADR TVVTIAHRVH TILSADLVMV LKRGAILEFD KPETLLSQKD SVFASFVRAD K UniProtKB:  SUR1 SUR1 |
-実験情報
-構造解析
| 手法 |  クライオ電子顕微鏡法 クライオ電子顕微鏡法 |
|---|---|
 解析 解析 |  単粒子再構成法 単粒子再構成法 |
| 試料の集合状態 | particle |
- 試料調製
試料調製
| 濃度 | 0.15 mg/mL | ||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 緩衝液 | pH: 7.4 構成要素:
詳細: HEPES, pH 7.5, 200 mM NaCl, 100 mM KCl, 0.05% digitonin, long-chain PIP2 | ||||||||||||||||||
| グリッド | モデル: Quantifoil R1.2/1.3 / 材質: GOLD / メッシュ: 300 / 支持フィルム - #0 - Film type ID: 1 / 支持フィルム - #0 - 材質: GRAPHENE OXIDE / 支持フィルム - #0 - トポロジー: CONTINUOUS / 支持フィルム - #1 - Film type ID: 2 / 支持フィルム - #1 - 材質: CARBON / 支持フィルム - #1 - トポロジー: HOLEY / 前処理 - タイプ: GLOW DISCHARGE / 前処理 - 時間: 45 sec. 詳細: Following glow-discharge, the grid was coated with Graphene Oxide before sample application | ||||||||||||||||||
| 凍結 | 凍結剤: ETHANE / チャンバー内湿度: 100 % / チャンバー内温度: 279 K / 装置: FEI VITROBOT MARK III | ||||||||||||||||||
| 詳細 | 3 microliters of purified Kir6.2-Q52R/FLAG-SUR1 were loaded onto Quantifoil R 1.2/1.3 Au 300 grids prepared with a fresh Graphene Oxide surface. |
- 電子顕微鏡法
電子顕微鏡法
| 顕微鏡 | FEI TITAN KRIOS |
|---|---|
| 電子線 | 加速電圧: 300 kV / 電子線源:  FIELD EMISSION GUN FIELD EMISSION GUN |
| 電子光学系 | C2レンズ絞り径: 70.0 µm / 照射モード: SPOT SCAN / 撮影モード: BRIGHT FIELD Bright-field microscopy / Cs: 2.7 mm / 最大 デフォーカス(公称値): 2.5 µm / 最小 デフォーカス(公称値): 1.0 µm / 倍率(公称値): 105000 Bright-field microscopy / Cs: 2.7 mm / 最大 デフォーカス(公称値): 2.5 µm / 最小 デフォーカス(公称値): 1.0 µm / 倍率(公称値): 105000 |
| 試料ステージ | 試料ホルダーモデル: FEI TITAN KRIOS AUTOGRID HOLDER ホルダー冷却材: NITROGEN |
| 詳細 | Titan Krios #3 at the Pacific Northwest National Lab |
| 撮影 | フィルム・検出器のモデル: GATAN K3 (6k x 4k) / 撮影したグリッド数: 3 / 実像数: 5241 / 平均露光時間: 2.2 sec. / 平均電子線量: 55.0 e/Å2 |
| 実験機器 |  モデル: Titan Krios / 画像提供: FEI Company |
- 画像解析
画像解析
| 粒子像選択 | 選択した数: 12320 / 詳細: C4 symmetry expanded particles |
|---|---|
| 初期モデル | モデルのタイプ: NONE |
| 初期 角度割当 | タイプ: MAXIMUM LIKELIHOOD / ソフトウェア - 名称: cryoSPARC (ver. 4.2.1) |
| 最終 3次元分類 | クラス数: 4 / ソフトウェア - 名称: cryoSPARC (ver. 4.2.1) 詳細: 3D classification parsed out a CTD-down closed conformation |
| 最終 角度割当 | タイプ: MAXIMUM LIKELIHOOD / ソフトウェア - 名称: cryoSPARC (ver. 4.2.1) |
| 最終 再構成 | 使用したクラス数: 1 / 想定した対称性 - 点群: C1 (非対称) / 解像度のタイプ: BY AUTHOR / 解像度: 6.9 Å / 解像度の算出法: FSC 0.143 CUT-OFF / ソフトウェア - 名称: cryoSPARC (ver. 4.2.1) 詳細: C4 symmetry expanded particles were used for the reconstruction 使用した粒子像数: 12320 |
-原子モデル構築 1
| 初期モデル | PDB ID: Chain - Source name: PDB / Chain - Initial model type: experimental model |
|---|---|
| 詳細 | A rigid-body fit of PDB ID 7UQR (closed apo Kir6.2/SUR1) is a good fit into this closed apo Kir6.2-Q52R/SUR1 map |
| 精密化 | プロトコル: RIGID BODY FIT |
 ムービー
ムービー コントローラー
コントローラー












 Z
Z Y
Y X
X