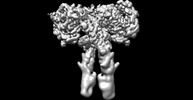
登録情報 データベース : EMDB / ID : EMD-43279タイトル Cryo-EM structure of short form insulin receptor (IR-A) with four IGF2 bound, symmetric conformation. Cryo-EM structure of short form insulin receptor (IR-A) with four IGF2 bound, symmetric conformation. 複合体 : Short form insulin receptor (IR-A) with four IGF2 bound, symmetric conformation.タンパク質・ペプチド : Isoform Short of Insulin receptorタンパク質・ペプチド : Insulin-like growth factor II / / / 機能・相同性 分子機能 ドメイン・相同性 構成要素
/ / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / / 生物種 Homo sapiens (ヒト)手法 / / 解像度 : 3.6 Å An W / Hall C / Li J / Huang A / Wu J / Park J / Bai XC / Choi E 資金援助 Organization Grant number 国 National Institutes of Health/National Institute of General Medical Sciences (NIH/NIGMS) GM136976
ジャーナル : Nat Commun / 年 : 2024タイトル : Activation of the insulin receptor by insulin-like growth factor 2.著者 : Weidong An / Catherine Hall / Jie Li / Albert Hung / Jiayi Wu / Junhee Park / Liwei Wang / Xiao-Chen Bai / Eunhee Choi / 要旨 : Insulin receptor (IR) controls growth and metabolism. Insulin-like growth factor 2 (IGF2) has different binding properties on two IR isoforms, mimicking insulin's function. However, the molecular ... Insulin receptor (IR) controls growth and metabolism. Insulin-like growth factor 2 (IGF2) has different binding properties on two IR isoforms, mimicking insulin's function. However, the molecular mechanism underlying IGF2-induced IR activation remains unclear. Here, we present cryo-EM structures of full-length human long isoform IR (IR-B) in both the inactive and IGF2-bound active states, and short isoform IR (IR-A) in the IGF2-bound active state. Under saturated IGF2 concentrations, both the IR-A and IR-B adopt predominantly asymmetric conformations with two or three IGF2s bound at site-1 and site-2, which differs from that insulin saturated IR forms an exclusively T-shaped symmetric conformation. IGF2 exhibits a relatively weak binding to IR site-2 compared to insulin, making it less potent in promoting full IR activation. Cell-based experiments validated the functional importance of IGF2 binding to two distinct binding sites in optimal IR signaling and trafficking. In the inactive state, the C-terminus of α-CT of IR-B contacts FnIII-2 domain of the same protomer, hindering its threading into the C-loop of IGF2, thus reducing the association rate of IGF2 with IR-B. Collectively, our studies demonstrate the activation mechanism of IR by IGF2 and reveal the molecular basis underlying the different affinity of IGF2 to IR-A and IR-B. 履歴 登録 2024年1月6日 - ヘッダ(付随情報) 公開 2024年3月27日 - マップ公開 2024年3月27日 - 更新 2024年4月3日 - 現状 2024年4月3日 処理サイト : RCSB / 状態 : 公開
すべて表示 表示を減らす
 データを開く
データを開く 基本情報
基本情報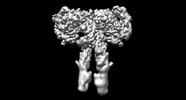
 マップデータ
マップデータ 試料
試料 キーワード
キーワード Insulin receptor (インスリン受容体) / IGF2 / RTK /
Insulin receptor (インスリン受容体) / IGF2 / RTK /  SIGNALING PROTEIN
SIGNALING PROTEIN 機能・相同性情報
機能・相同性情報 ゲノム刷り込み ...spongiotrophoblast cell proliferation / positive regulation of skeletal muscle tissue growth / negative regulation of muscle cell differentiation / embryonic placenta morphogenesis / regulation of muscle cell differentiation / Signaling by Type 1 Insulin-like Growth Factor 1 Receptor (IGF1R) / IRS-related events triggered by IGF1R / regulation of female gonad development / positive regulation of meiotic cell cycle /
ゲノム刷り込み ...spongiotrophoblast cell proliferation / positive regulation of skeletal muscle tissue growth / negative regulation of muscle cell differentiation / embryonic placenta morphogenesis / regulation of muscle cell differentiation / Signaling by Type 1 Insulin-like Growth Factor 1 Receptor (IGF1R) / IRS-related events triggered by IGF1R / regulation of female gonad development / positive regulation of meiotic cell cycle /  ゲノム刷り込み / positive regulation of organ growth / positive regulation of developmental growth /
ゲノム刷り込み / positive regulation of organ growth / positive regulation of developmental growth /  insulin-like growth factor II binding / male sex determination / exocrine pancreas development /
insulin-like growth factor II binding / male sex determination / exocrine pancreas development /  insulin receptor complex /
insulin receptor complex /  insulin-like growth factor I binding / positive regulation of multicellular organism growth /
insulin-like growth factor I binding / positive regulation of multicellular organism growth /  insulin receptor activity / positive regulation of protein-containing complex disassembly / cargo receptor activity / positive regulation of vascular endothelial cell proliferation /
insulin receptor activity / positive regulation of protein-containing complex disassembly / cargo receptor activity / positive regulation of vascular endothelial cell proliferation /  dendritic spine maintenance /
dendritic spine maintenance /  insulin binding /
insulin binding /  PTB domain binding / adrenal gland development / neuronal cell body membrane / Signaling by Insulin receptor /
PTB domain binding / adrenal gland development / neuronal cell body membrane / Signaling by Insulin receptor /  transmembrane receptor protein tyrosine kinase activator activity / IRS activation / positive regulation of activated T cell proliferation / amyloid-beta clearance / activation of protein kinase activity / positive regulation of respiratory burst /
transmembrane receptor protein tyrosine kinase activator activity / IRS activation / positive regulation of activated T cell proliferation / amyloid-beta clearance / activation of protein kinase activity / positive regulation of respiratory burst /  regulation of embryonic development / positive regulation of receptor internalization / transport across blood-brain barrier / positive regulation of cell division /
regulation of embryonic development / positive regulation of receptor internalization / transport across blood-brain barrier / positive regulation of cell division /  insulin receptor substrate binding / embryonic placenta development / epidermis development / positive regulation of glycogen biosynthetic process / SHC-related events triggered by IGF1R / Signal attenuation /
insulin receptor substrate binding / embryonic placenta development / epidermis development / positive regulation of glycogen biosynthetic process / SHC-related events triggered by IGF1R / Signal attenuation /  phosphatidylinositol 3-kinase binding / heart morphogenesis / positive regulation of insulin receptor signaling pathway / dendrite membrane / striated muscle cell differentiation / Insulin receptor recycling /
phosphatidylinositol 3-kinase binding / heart morphogenesis / positive regulation of insulin receptor signaling pathway / dendrite membrane / striated muscle cell differentiation / Insulin receptor recycling /  insulin-like growth factor receptor binding / neuron projection maintenance / activation of protein kinase B activity / protein serine/threonine kinase activator activity / positive regulation of glycolytic process / Insulin receptor signalling cascade /
insulin-like growth factor receptor binding / neuron projection maintenance / activation of protein kinase B activity / protein serine/threonine kinase activator activity / positive regulation of glycolytic process / Insulin receptor signalling cascade /  receptor-mediated endocytosis / positive regulation of mitotic nuclear division / insulin-like growth factor receptor signaling pathway / platelet alpha granule lumen /
receptor-mediated endocytosis / positive regulation of mitotic nuclear division / insulin-like growth factor receptor signaling pathway / platelet alpha granule lumen /  学習 /
学習 /  カベオラ / positive regulation of glucose import / animal organ morphogenesis /
カベオラ / positive regulation of glucose import / animal organ morphogenesis /  growth factor activity / positive regulation of MAP kinase activity /
growth factor activity / positive regulation of MAP kinase activity /  insulin receptor binding /
insulin receptor binding /  hormone activity /
hormone activity /  receptor internalization /
receptor internalization /  受容体型チロシンキナーゼ /
受容体型チロシンキナーゼ /  記憶 / cellular response to growth factor stimulus / osteoblast differentiation / peptidyl-tyrosine phosphorylation / cellular response to insulin stimulus / male gonad development /
記憶 / cellular response to growth factor stimulus / osteoblast differentiation / peptidyl-tyrosine phosphorylation / cellular response to insulin stimulus / male gonad development /  cell surface receptor protein tyrosine kinase signaling pathway / glucose metabolic process / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / positive regulation of nitric oxide biosynthetic process / positive regulation of peptidyl-tyrosine phosphorylation /
cell surface receptor protein tyrosine kinase signaling pathway / glucose metabolic process / Regulation of Insulin-like Growth Factor (IGF) transport and uptake by Insulin-like Growth Factor Binding Proteins (IGFBPs) / positive regulation of nitric oxide biosynthetic process / positive regulation of peptidyl-tyrosine phosphorylation /  integrin binding / late endosome /
integrin binding / late endosome /  glucose homeostasis / Platelet degranulation / insulin receptor signaling pathway /
glucose homeostasis / Platelet degranulation / insulin receptor signaling pathway /  amyloid-beta binding / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling /
amyloid-beta binding / PI5P, PP2A and IER3 Regulate PI3K/AKT Signaling /  protein tyrosine kinase activity / in utero embryonic development / positive regulation of MAPK cascade / protein autophosphorylation /
protein tyrosine kinase activity / in utero embryonic development / positive regulation of MAPK cascade / protein autophosphorylation /  リソソーム /
リソソーム /  receptor ligand activity / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction /
receptor ligand activity / positive regulation of phosphatidylinositol 3-kinase/protein kinase B signal transduction /  receptor complex / endosome membrane / carbohydrate metabolic process / positive regulation of cell migration / positive regulation of protein phosphorylation
receptor complex / endosome membrane / carbohydrate metabolic process / positive regulation of cell migration / positive regulation of protein phosphorylation
 Homo sapiens (ヒト)
Homo sapiens (ヒト) 単粒子再構成法 /
単粒子再構成法 /  クライオ電子顕微鏡法 / 解像度: 3.6 Å
クライオ電子顕微鏡法 / 解像度: 3.6 Å  データ登録者
データ登録者 米国, 1件
米国, 1件  引用
引用 ジャーナル: Nat Commun / 年: 2024
ジャーナル: Nat Commun / 年: 2024
 構造の表示
構造の表示 ダウンロードとリンク
ダウンロードとリンク emd_43279.map.gz
emd_43279.map.gz EMDBマップデータ形式
EMDBマップデータ形式 emd-43279-v30.xml
emd-43279-v30.xml emd-43279.xml
emd-43279.xml EMDBヘッダ
EMDBヘッダ emd_43279.png
emd_43279.png emd-43279.cif.gz
emd-43279.cif.gz emd_43279_half_map_1.map.gz
emd_43279_half_map_1.map.gz emd_43279_half_map_2.map.gz
emd_43279_half_map_2.map.gz http://ftp.pdbj.org/pub/emdb/structures/EMD-43279
http://ftp.pdbj.org/pub/emdb/structures/EMD-43279 ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43279
ftp://ftp.pdbj.org/pub/emdb/structures/EMD-43279 リンク
リンク EMDB (EBI/PDBe) /
EMDB (EBI/PDBe) /  EMDataResource
EMDataResource マップ
マップ ダウンロード / ファイル: emd_43279.map.gz / 形式: CCP4 / 大きさ: 75.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES)
ダウンロード / ファイル: emd_43279.map.gz / 形式: CCP4 / 大きさ: 75.1 MB / タイプ: IMAGE STORED AS FLOATING POINT NUMBER (4 BYTES) 試料の構成要素
試料の構成要素
 Homo sapiens (ヒト)
Homo sapiens (ヒト) 受容体型チロシンキナーゼ
受容体型チロシンキナーゼ
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Homo sapiens (ヒト)
Homo sapiens (ヒト) インスリン受容体
インスリン受容体
 Homo sapiens (ヒト)
Homo sapiens (ヒト)
 Escherichia coli (大腸菌)
Escherichia coli (大腸菌) インスリン様成長因子2
インスリン様成長因子2 クライオ電子顕微鏡法
クライオ電子顕微鏡法 解析
解析 単粒子再構成法
単粒子再構成法 試料調製
試料調製 電子顕微鏡法
電子顕微鏡法 FIELD EMISSION GUN
FIELD EMISSION GUN Bright-field microscopy / 最大 デフォーカス(公称値): 2.6 µm / 最小 デフォーカス(公称値): 1.6 µm
Bright-field microscopy / 最大 デフォーカス(公称値): 2.6 µm / 最小 デフォーカス(公称値): 1.6 µm
 画像解析
画像解析 ) / 解像度のタイプ: BY AUTHOR / 解像度: 3.6 Å / 解像度の算出法: FSC 0.143 CUT-OFF / ソフトウェア - 名称: RELION / 使用した粒子像数: 66937
) / 解像度のタイプ: BY AUTHOR / 解像度: 3.6 Å / 解像度の算出法: FSC 0.143 CUT-OFF / ソフトウェア - 名称: RELION / 使用した粒子像数: 66937 
 ムービー
ムービー コントローラー
コントローラー




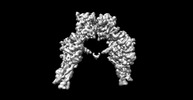


















 Z
Z Y
Y X
X