+Search query
-Structure paper
| Title | Structural basis and synergism of ATP and Na activation in bacterial K uptake system KtrAB. |
|---|---|
| Journal, issue, pages | Nat Commun, Vol. 15, Issue 1, Page 3850, Year 2024 |
| Publish date | May 8, 2024 |
 Authors Authors | Wesley Tien Chiang / Yao-Kai Chang / Wei-Han Hui / Shu-Wei Chang / Chen-Yi Liao / Yi-Chuan Chang / Chun-Jung Chen / Wei-Chen Wang / Chien-Chen Lai / Chun-Hsiung Wang / Siou-Ying Luo / Ya-Ping Huang / Shan-Ho Chou / Tzyy-Leng Horng / Ming-Hon Hou / Stephen P Muench / Ren-Shiang Chen / Ming-Daw Tsai / Nien-Jen Hu /   |
| PubMed Abstract | The K uptake system KtrAB is essential for bacterial survival in low K environments. The activity of KtrAB is regulated by nucleotides and Na. Previous studies proposed a putative gating mechanism of ...The K uptake system KtrAB is essential for bacterial survival in low K environments. The activity of KtrAB is regulated by nucleotides and Na. Previous studies proposed a putative gating mechanism of KtrB regulated by KtrA upon binding to ATP or ADP. However, how Na activates KtrAB and the Na binding site remain unknown. Here we present the cryo-EM structures of ATP- and ADP-bound KtrAB from Bacillus subtilis (BsKtrAB) both solved at 2.8 Å. A cryo-EM density at the intra-dimer interface of ATP-KtrA was identified as Na, as supported by X-ray crystallography and ICP-MS. Thermostability assays and functional studies demonstrated that Na binding stabilizes the ATP-bound BsKtrAB complex and enhances its K flux activity. Comparing ATP- and ADP-BsKtrAB structures suggests that BsKtrB Arg417 and Phe91 serve as a channel gate. The synergism of ATP and Na in activating BsKtrAB is likely applicable to Na-activated K channels in central nervous system. |
 External links External links |  Nat Commun / Nat Commun /  PubMed:38719864 / PubMed:38719864 /  PubMed Central PubMed Central |
| Methods | EM (single particle) / X-ray diffraction |
| Resolution | 2.48 - 3.1 Å |
| Structure data | EMDB-36800, PDB-8k1s:  EMDB-36801: Potassium transporter KtrAB from Bacillus subtilis in ADP-bound state, focused refined on KtrA octamer  EMDB-36802: Potassium transporter KtrAB from Bacillus subtilis in ADP-bound state, focused refined on KtrB dimer EMDB-36803, PDB-8k1t: EMDB-36804, PDB-8k1u: EMDB-38477, PDB-8xmh: EMDB-38478, PDB-8xmi:  PDB-8k16:  PDB-8k1k: |
| Chemicals | 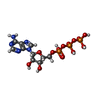 ChemComp-ATP:  ChemComp-TL:  ChemComp-NA: 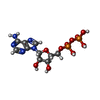 ChemComp-ADP:  ChemComp-K: |
| Source |
|
 Keywords Keywords | METAL TRANSPORT / potassium channel RCK domain / METAL BINDING PROTEIN /  Potassium channel / Potassium channel /  TRANSPORT PROTEIN / KtrAB / RCK / TRANSPORT PROTEIN / KtrAB / RCK /  potassium / potassium /  transporter transporter |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers














