+Search query
-Structure paper
| Title | Structure of the native γ-tubulin ring complex capping spindle microtubules. |
|---|---|
| Journal, issue, pages | Nat Struct Mol Biol, Year 2024 |
| Publish date | Apr 12, 2024 |
 Authors Authors | Tom Dendooven / Stanislau Yatskevich / Alister Burt / Zhuo A Chen / Dom Bellini / Juri Rappsilber / John V Kilmartin / David Barford /    |
| PubMed Abstract | Microtubule (MT) filaments, composed of α/β-tubulin dimers, are fundamental to cellular architecture, function and organismal development. They are nucleated from MT organizing centers by the ...Microtubule (MT) filaments, composed of α/β-tubulin dimers, are fundamental to cellular architecture, function and organismal development. They are nucleated from MT organizing centers by the evolutionarily conserved γ-tubulin ring complex (γTuRC). However, the molecular mechanism of nucleation remains elusive. Here we used cryo-electron tomography to determine the structure of the native γTuRC capping the minus end of a MT in the context of enriched budding yeast spindles. In our structure, γTuRC presents a ring of γ-tubulin subunits to seed nucleation of exclusively 13-protofilament MTs, adopting an active closed conformation to function as a perfect geometric template for MT nucleation. Our cryo-electron tomography reconstruction revealed that a coiled-coil protein staples the first row of α/β-tubulin of the MT to alternating positions along the γ-tubulin ring of γTuRC. This positioning of α/β-tubulin onto γTuRC suggests a role for the coiled-coil protein in augmenting γTuRC-mediated MT nucleation. Based on our results, we describe a molecular model for budding yeast γTuRC activation and MT nucleation. |
 External links External links |  Nat Struct Mol Biol / Nat Struct Mol Biol /  PubMed:38609662 PubMed:38609662 |
| Methods | EM (subtomogram averaging) |
| Resolution | 6.6 - 9.2 Å |
| Structure data | EMDB-18664, PDB-8qv0: EMDB-18665, PDB-8qv2: EMDB-18666, PDB-8qv3: |
| Chemicals | 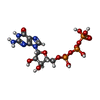 ChemComp-GTP: 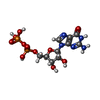 ChemComp-GDP:  ChemComp-HOH: |
| Source |
|
 Keywords Keywords |  CELL CYCLE / CELL CYCLE /  Microtubule nucleation / MTOC / y-tubulin / SPB Microtubule nucleation / MTOC / y-tubulin / SPB |
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers










