+検索条件
-Structure paper
| タイトル | Postcatalytic spliceosome structure reveals mechanism of 3'-splice site selection. |
|---|---|
| ジャーナル・号・ページ | Science, Vol. 358, Issue 6368, Page 1283-1288, Year 2017 |
| 掲載日 | 2017年12月8日 |
 著者 著者 | Max E Wilkinson / Sebastian M Fica / Wojciech P Galej / Christine M Norman / Andrew J Newman / Kiyoshi Nagai /  |
| PubMed 要旨 | Introns are removed from eukaryotic messenger RNA precursors by the spliceosome in two transesterification reactions-branching and exon ligation. The mechanism of 3'-splice site recognition during ...Introns are removed from eukaryotic messenger RNA precursors by the spliceosome in two transesterification reactions-branching and exon ligation. The mechanism of 3'-splice site recognition during exon ligation has remained unclear. Here we present the 3.7-angstrom cryo-electron microscopy structure of the yeast P-complex spliceosome immediately after exon ligation. The 3'-splice site AG dinucleotide is recognized through non-Watson-Crick pairing with the 5' splice site and the branch-point adenosine. After the branching reaction, protein factors work together to remodel the spliceosome and stabilize a conformation competent for 3'-splice site docking, thereby promoting exon ligation. The structure accounts for the strict conservation of the GU and AG dinucleotides at the 5' and 3' ends of introns and provides insight into the catalytic mechanism of exon ligation. |
 リンク リンク |  Science / Science /  PubMed:29146871 / PubMed:29146871 /  PubMed Central PubMed Central |
| 手法 | EM (単粒子) |
| 解像度 | 3.7 - 4.0 Å |
| 構造データ | EMDB-3979, PDB-6exn:  EMDB-3980: |
| 化合物 |  ChemComp-MG: 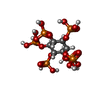 ChemComp-IHP: 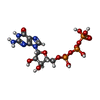 ChemComp-GTP:  ChemComp-ZN: |
| 由来 |
|
 キーワード キーワード |  SPLICING / SPLICING /  Spliceosome (スプライセオソーム) / P-complex / Exon ligation Spliceosome (スプライセオソーム) / P-complex / Exon ligation |
 ムービー
ムービー コントローラー
コントローラー 構造ビューア
構造ビューア 万見文献について
万見文献について






