+Search query
-Structure paper
| Title | Polymorphism and double hexamer structure in the archaeal minichromosome maintenance (MCM) helicase from Methanobacterium thermoautotrophicum. |
|---|---|
| Journal, issue, pages | J Biol Chem, Vol. 280, Issue 49, Page 40909-40915, Year 2005 |
| Publish date | Dec 9, 2005 |
 Authors Authors | Yacob Gómez-Llorente / Ryan J Fletcher / Xiaojiang S Chen / José M Carazo / Carmen San Martín /  |
| PubMed Abstract | Methanobacterium thermoautotrophicum minichromosome maintenance complex (mtMCM), a cellular replicative helicase, is a useful model for the more complex eukaryotic MCMs. Biochemical and ...Methanobacterium thermoautotrophicum minichromosome maintenance complex (mtMCM), a cellular replicative helicase, is a useful model for the more complex eukaryotic MCMs. Biochemical and crystallographic evidence indicates that mtMCM assembles as a double hexamer (dHex), but previous electron microscopy studies reported only the presence of single heptamers or single hexamers (Pape, T., Meka, H., Chen, S., Vicentini, G., Van Heel, M., and Onesti, S. (2003) EMBO Rep. 4, 1079-1083; Yu, X., VanLoock, M. S., Poplawski, A., Kelman, Z., Xiang, T., Tye, B. K., and Egelman, E. H. (2002) EMBO Rep. 3, 792-797). Here we present the first three-dimensional electron microscopy reconstruction of the full-length mtMCM dHex in which two hexamers contact each other via the structurally well defined N-terminal domains. The dHex has obvious side openings that resemble the side channels of LTag (large T antigen). 6-fold and 7-fold rings were observed in the same mtMCM preparation, but we determined that assembly as a double ring favors 6-fold structures. Additionally, open rings were also detected, which suggests a direct mtMCM loading mechanism onto DNA. |
 External links External links |  J Biol Chem / J Biol Chem /  PubMed:16221680 PubMed:16221680 |
| Methods | EM (single particle) |
| Resolution | 25.0 Å |
| Structure data | 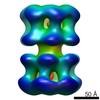 EMDB-1134: |
| Source |
|
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers





 Methanothermobacter thermautotrophicus (archaea)
Methanothermobacter thermautotrophicus (archaea)