+Search query
-Structure paper
| Title | ATP-driven remodeling of the linker domain in the dynein motor. |
|---|---|
| Journal, issue, pages | Structure, Vol. 20, Issue 10, Page 1670-1680, Year 2012 |
| Publish date | Oct 10, 2012 |
 Authors Authors | Anthony J Roberts / Bara Malkova / Matt L Walker / Hitoshi Sakakibara / Naoki Numata / Takahide Kon / Reiko Ohkura / Thomas A Edwards / Peter J Knight / Kazuo Sutoh / Kazuhiro Oiwa / Stan A Burgess /  |
| PubMed Abstract | Dynein ATPases are the largest known cytoskeletal motors and perform critical functions in cells: carrying cargo along microtubules in the cytoplasm and powering flagellar beating. Dyneins are ...Dynein ATPases are the largest known cytoskeletal motors and perform critical functions in cells: carrying cargo along microtubules in the cytoplasm and powering flagellar beating. Dyneins are members of the AAA+ superfamily of ring-shaped enzymes, but how they harness this architecture to produce movement is poorly understood. Here, we have used cryo-EM to determine 3D maps of native flagellar dynein-c and a cytoplasmic dynein motor domain in different nucleotide states. The structures show key sites of conformational change within the AAA+ ring and a large rearrangement of the "linker" domain, involving a hinge near its middle. Analysis of a mutant in which the linker "undocks" from the ring indicates that linker remodeling requires energy that is supplied by interactions with the AAA+ modules. Fitting the dynein-c structures into flagellar tomograms suggests how this mechanism could drive sliding between microtubules, and also has implications for cytoplasmic cargo transport. |
 External links External links |  Structure / Structure /  PubMed:22863569 / PubMed:22863569 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 19.0 - 22.0 Å |
| Structure data | 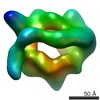 EMDB-2155: 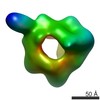 EMDB-2156: |
| Source |
|
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers




 Chlamydomonas reinhardtii (plant)
Chlamydomonas reinhardtii (plant)