+Search query
-Structure paper
| Title | The eye lens chaperone alpha-crystallin forms defined globular assemblies. |
|---|---|
| Journal, issue, pages | Proc Natl Acad Sci U S A, Vol. 106, Issue 32, Page 13272-13277, Year 2009 |
| Publish date | Aug 11, 2009 |
 Authors Authors | Jirka Peschek / Nathalie Braun / Titus M Franzmann / Yannis Georgalis / Martin Haslbeck / Sevil Weinkauf / Johannes Buchner /  |
| PubMed Abstract | Alpha-crystallins are molecular chaperones that protect vertebrate eye lens proteins from detrimental protein aggregation. alphaB-Crystallin, 1 of the 2 alpha-crystallin isoforms, is also associated ...Alpha-crystallins are molecular chaperones that protect vertebrate eye lens proteins from detrimental protein aggregation. alphaB-Crystallin, 1 of the 2 alpha-crystallin isoforms, is also associated with myopathies and neuropathological diseases. Despite the importance of alpha-crystallins in protein homeostasis, only little is known about their quaternary structures because of their seemingly polydisperse nature. Here, we analyzed the structures of recombinant alpha-crystallins using biophysical methods. In contrast to previous reports, we show that alphaB-crystallin assembles into defined oligomers consisting of 24 subunits. The 3-dimensional (3D) reconstruction of alphaB-crystallin by electron microscopy reveals a sphere-like structure with large openings to the interior of the protein. alphaA-Crystallin forms, in addition to complexes of 24 subunits, also smaller oligomers and large clusters consisting of individual oligomers. This propensity might explain the previously reported polydisperse nature of alpha-crystallin. |
 External links External links |  Proc Natl Acad Sci U S A / Proc Natl Acad Sci U S A /  PubMed:19651604 / PubMed:19651604 /  PubMed Central PubMed Central |
| Methods | EM (single particle) |
| Resolution | 20.0 Å |
| Structure data | 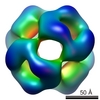 EMDB-1776: |
| Source |
|
 Movie
Movie Controller
Controller Structure viewers
Structure viewers About Yorodumi Papers
About Yorodumi Papers




 Homo sapiens (human)
Homo sapiens (human)